Draw the major product(s) that are expected when each of the following amines is treated with excess
Question:
(a)
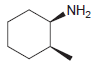
(b)
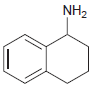
Transcribed Image Text:
NH2 NH2
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 85% (14 reviews)

Answered By

Vincent Omondi
I am an extremely self-motivated person who firmly believes in his abilities. With high sensitivity to task and operating parameters, deadlines and keen on instructions, I deliver the best quality work for my clients. I handle tasks ranging from assignments to projects.
4.90+
109+ Reviews
314+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
Predict the products that are expected when each of the following alkenes is treated with a peroxy acid (such as MCPBA) followed by aqueous acid: a. b. c. d. e. f.
-
Predict the products that are expected when each of the following alkenes is treated with ozone followed by DMS: a. b. c. d. e. f.
-
What product(s) are expected when each of the following compounds reacts with one equivalent of NBS in CC14 in the presence of light and peroxides? Explain your answers. (a) cyclohexene (b)...
-
In Fig. P2.72 gate AB is circular. Find the moment of the hydrostatic force on this gate about axis A. Neglect atmospheric pressure.
-
Find the sum of the finite geometric sequence. 1. 2. 3. 4. -1 2i i=1 31 i=1
-
At the beginning of 2017, Holden Companys controller asked you to prepare correcting entries for the following three situations: 1. Machine X was purchased for $100,000 on January 1, 2012....
-
The life span (in kilometers) for a population of a new brand of tires can be approximated by a normal distribution, with a mean of 80,000 kilometers and a standard deviation of 1,500 kilometers. (a)...
-
Complete the profit margin, asset turnover, and return on investment calculations for investment centers D and V. (Round to two decimalplaces.) Center D $1,600 $400 $3,200 Center V $2,000 $200 $1,250...
-
1) Leadership and management are very important vital components to an effective functioning organization so that people in the organization are inspired, managed and guided towards a common vision....
-
After looking at the projections of the HomeNet project, you decide that they are not realistic. It is unlikely that sales will be constant over the four-year life of the project. Furthermore, other...
-
Consider the structure of the azo dye called alizarine yellow R (below). Show the reagents you would use to prepare this compound via an azo coupling process. .N. N' O2N
-
If Davis adds a line of apparel or shoes, should he try to sell his line to other retail clothing shops in Calgary or elsewhere in Canada?
-
Name the classes to which the following compounds belong: (a) C4H9OH (b) CH3OC2H5 (c) C2H5CHO (d) C6H5COOH (e) CH3NH2
-
Advocates must do background research to understand what decision makers care about and what could influence them. Ask yourself the following questions: What motivates your key decision makers? Who...
-
What do you think of the term medical error as a descriptor of adverse medical outcomes? After all, there are many medical procedures (e.g., invasive surgeries) and treatments (e.g., chemotherapy)...
-
Congress passed the various pandemic laws without paying for them. The federal deficit is projected to be $2.3 trillion in 2021, equaling 10.3% of GDP. This is the second largest deficit since 1945,...
-
Are there alternatives to the individual mandate that accomplish the same goals without engendering so much political turmoil? Could policymakers have designed an incentive system that would be as...
-
While Democrats passed the ACA and remain its strongest supporters, not all Democrats and progressive constituents believe the ACAs approach was the best way to reform the healthcare system. During...
-
Determine the different possibilities for the numbers of positive, negative, and nonreal complex zeros of each function. (x) = 11x 5 - x 3 + 7x - 5
-
What are the 5 Cs of marketing channel structure?
-
Predict which compound in each pair has the higher boiling point. Explain your prediction. (a) CH3CH2OCH3 or CH3CH1OH2CH3 (b) CH3CH2CH2CH3 or CH3CH2CH2CH2CH3 (c) CH3CH2CH2CH2CH3 or (CH3)2CHCH2CH3 (d)...
-
Circle the functional groups in the following structures. State to which class (or classes) of compounds the structure belongs. (a) (b) (c) (d) (e) (f) (g) (h) (i) COOH N--H CH2OCH3 CN NH CH3 CHCOOCH...
-
Dimethyl sulfoxide (DMSO) has been used as an anti-inflammatory rub for race horses. DMSO and acetone appear to have similar structures, but the C=O carbon atom in acetone is planar, while the S=O...
-
Question: Read the document on this link: https://pui2022q4.tiiny.site/ 1. You form part of a company in South Africa and you have to continuously do business locally and internationally on the...
-
Banjo Education Corp. issued a 4%, $80,000 bond that pays interest semiannually each June 30 and December 31. The date of issuance was January 1, 2020. The bonds mature after four years. The market...
-
A projectile if fired from 19.6m above the ground with an initial speed of 55.6m/s at an angle of 41.2 degrees above the horizontal. a) Determine the time necessary for the projectile to reach its...

Study smarter with the SolutionInn App


