Give the structure of the alkene that could be used as a starting material to form chlorohydrin
Question:
Give the structure of the alkene that could be used as a starting material to form chlorohydrin B in Study Problem 5.1.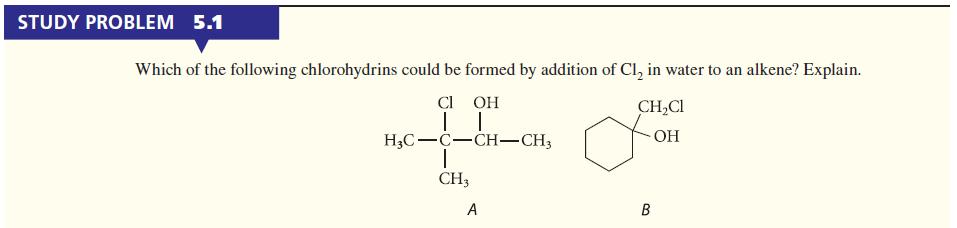
Transcribed Image Text:
STUDY PROBLEM 5.1 Which of the following chlorohydrins could be formed by addition of Cl₂ in water to an alkene? Explain. CI OH TI H3C-C-CH-CH3 1 CH3 A CH₂Cl OH B
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 50% (2 reviews)
The alkene that could ...View the full answer

Answered By

BRIAN MUSINGA
I possess a Bachelors of Commerce degree(Marketing option) and am currently undertaking an MBA in marketing. I believe that I possess the required knowledge and skills to tutor in the subject named. I have also written numerous research academic papers much to the satisfaction of clients and my professors.
5.00+
2+ Reviews
17+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
Write a project management plan. we have a template and project description. we need to edit the template(table of contents) with our own ideas. CPSC 8820-01 Project Management Plan Your Unique...
-
A study was conducted on a sample of 53 patients presenting with prostate cancer who had also undergone a laparotomy to ascertain the extent of nodal involvement (Collett 1991). The result of the...
-
Give two sets of alkyl bromide and alkene that could be used in a Heck reaction to prepare the following compound: CH3C CH= CH OCH3
-
Explain why merchants accepted gold receipts as a means of payment even though the receipts were issued by gold-smiths, not the government. What risk did goldsmiths introduce into the payments system...
-
Multiple-Choice Questions 1. In a make-or-buy decision, a. The company must choose between expanding or dropping a product line. b. The company must choose between accepting or rejecting a special...
-
Sussman Industries purchased a drilling machine for $50,000 and paid cash. Sussman expects to use the machine for ten years after which it will have no value. It will be depreciated straight-line...
-
Use the same facts as problem 20 and prepare entries using the following independent assumptions: a. Lease A is abandoned in 2022. b. Lease A is proved in 2022. c. Insignificant Lease Y, with a cost...
-
The financial results for the past two years for Ornamental Iron, a division of Iron Foundry Company, follow: Required 1. Compute the division's profit margin, asset turnover, and return on...
-
Find the distance 'x' in this figure, accurate to 3 decimal places (i.e. 0.001"). (Hint: Locate two important points to form the required right-triangle to solve the question. The circle center is...
-
Alkenes undergo the addition of thiols at high temperature in the presence of peroxides or other free-radical initiators. The following reaction is an example. Propose a mechanism for this reaction....
-
Alkene X of unknown structure gives the following products after treatment with ozone followed by aqueous H 2 O 2 : What is the structure of X? =0 cyclopentanone and HCCH,CH3 propionic acid
-
Jacaranda Builders is undertaking an analysis of supplier costs in order to evaluate the relative costs and performance of each supplier. They have isolated a range of activities that are consumed by...
-
A partner has contributed property and its associated liability to a partnership in exchange for a partnership interest. What is the amount that the partnership will record as tax basis of the...
-
Briefly describe how hemoglobin and the carbonic acid buffering system work together in extrapulmonary tissues to buffer the blood and transport the blood gases.
-
The current spot price of gold is $1280 per ounce. The forward price for 1 year delivery is $1400. Suppose an investor can borrow at 3% per year (simple interest). How can the investor make a...
-
You are a senior project finance account manager at a boutique investment bank who has received the attached summarized project information memorandum ("Development, Financing and Operation of a 5MW...
-
Calculate the overtime pay. reference cell c21 for the "hours worked". the employee is paid 1.5 times the regular pay rate for any time the employee works more than the number regular hours in pay...
-
Portions of the financial statements for Parnell Company are provided below. Required: 1. Prepare the cash flows from operating activities section of the statement of cash flows for Parnell Company...
-
The time to assemble the first unit on a production line is 10 hours. The learning rate is 0.94. Approximately how long will it take for the seventh unit to be assembled? The number of hours needed...
-
Name these compounds: a) CHCH,CH,CHCH,CH,CH3 CHCH3 T CH3 b)
-
Draw structures for these compounds. (a) 4-(1-Methylethl) heptanes (b) 3-Ethyl-7-methyl-5-(1-methylpropyl) undecane
-
Designate each carbon of these compounds as primary, secondary, tertiary, or quaternary: CH3 CH3 CH a) CH,CCHCH,CH, CH3 c) 2,3-Dimethylpentane b) t
-
A spacecraft is designed so that it can accelerate in both the x and y directions with one rocket pointing toward the -x direction (left) and one rocket pointing toward the + y direction. The rocket...
-
The picture shows the initial velocity vectors for two particles that start at the origin. The velocity vectors are drawn to scale - you can assume the tip of each vector is on a a grid intersection...
-
4. The World Solar Challenge in 1987 was the first car race in which all the vehicles were solar powered. The winner was the GM Sunraycer, which had a mass of 177.4 kg, not counting the driver's...

Study smarter with the SolutionInn App


