In each of the following series, arrange the compounds in order of increasing reactivity in the E2
Question:
In each of the following series, arrange the compounds in order of increasing reactivity in the E2 reaction with Na+ EtO–.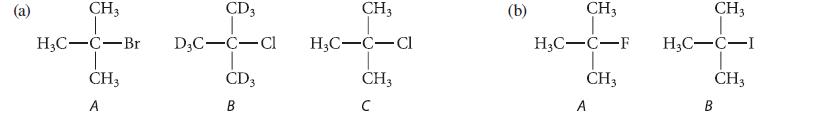
Transcribed Image Text:
CH3 H₂C-C-Br CH3 A CD3 D3C-C-Cl T CD3 B CH3 H₂C-C-Cl CH3 C (b) CH3 H3C-C-F CH3 A CH3 H₂C-C-I CH3 B
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 100% (QA)
a A primary deuterium isotope effect retards the reac...View the full answer

Answered By

IPPILI SUDHEER KUMAR
i done M TECH at jntu kakinada university i am second rank in my university and i have five years experience in teaching
0.00
0 Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
Arrange the compounds in each of the following groups in order of increasing solubility in water, and briefly explain your answers: a. 1-octanol; ethanol; ethyl chloride b. HOCH2(CHOH)3CH2OH;...
-
1) As a result of the US Senate elections in Georgia on January 5, 2021, Democrats now hold majority in both chambers of the US congress, which will likely lead to a significant increase in...
-
In each of the following sets, rank the compounds in order of increasing harshness of the reaction conditions required to accomplish the indicated reaction. Friedel-Crafts acylation of chlorobenzene,...
-
1. Given the image of the circle, identify the length of the radius, diameter, and circumference. Also, find the area. Make sure to show work (area and circumference) to receive credit. Choose a...
-
On January 1, 2012, the first day of its fiscal year, the City of Carter received notification that a federal grant in the amount of $650,000 was approved. The grant was restricted for the payment of...
-
1. What are key elements of country risk in Zimbabwe? 2. How has increased country risk affected Zimbabwes economy and living standards? 3. By how much is the Zim dollar at its official rate...
-
The cash flows associated with a project are shown below. The interest rate varies from year to year as shown. Determine an equivalent uniform annual series of cash flows. EOY Cash Flow Interest...
-
The Walton Toy Company manufactures a line of dolls and a doll dress sewing kit. Demand for the dolls is increasing, and management requests assistance from you in determining an economical sales and...
-
The December 3 1 , 2 0 2 4 , adjusted trial balance for the Blueboy Cheese Corporation is presented below. Account Title Debits Credits Cash $ 2 1 , 0 0 0 Accounts receivable 3 0 0 , 0 0 0 Prepaid...
-
What nucleophile or base and what type of solvent could be used for the conversion of isobutyl bromide into each of the following compounds? (a) (CH3)CHCHS (CH3)2 Br (b) (CH3)CHCHSCHCH3 (c)...
-
Which nucleophile, ;N(C 2 H 5 ) 3 or ;P(C 2 H 5 ) 3 , reacts most rapidly with methyl iodide in ethanol solvent? Explain, and give the product formed in each case.
-
(a) Find the approximations T10 and S10 for 1 ex and the corresponding errors ET and ES. (b) Compare the actual errors in part (a) with the error estimates given by (3) and (4). (c) How large do we...
-
Suppose that Ally Financial Inc. issued a bond with 10 years until maturity, a face value of $1000, and a coupon rate of 11% (annual payments). The yield to maturity on this bond when it was issued...
-
Members of international alliances often leave or change alliances. What factors cause this instability?
-
Can fuel hedging lower fuel costs? Why do some airlines hedge fuel prices?
-
Suppose you purchase a 10-year bond with 11% annual coupons. You hold the bond for four years, and sell it immediately after receiving the fourth coupon. If the bonds yield to maturity was 9.94% when...
-
What are the various ways that a state- owned airline can be privatized?
-
Given the following information, calculate the amount of after-tax profit for the period: sales, $260; expenses other than depreciation, $140; depreciation, $50; marginal income tax rate, 35 percent....
-
A statistical study shows that the fraction of television sets of a certain brand that are still in service after x years is given by f (x) = e-0.15x. (a) What fraction of the sets are still in...
-
What products would you expect from reaction of the following alkenes with NBS? If more than one product is formed, show the structures ofall. H (a) . (b) CH3D2H
-
How would you prepare the following alkyl halides from the correspondingalcohols? CH (a) CI (b) Br CH CHH2CH CH (c) CI H (d) BrCH2CH2CH2CHCH CHCH2H2cH3 CH
-
How strong a base would you expect a Grignard reagent to be? Look at Table 8.1, and then predict whether the following reactions will occur as written.?(The p K a of NH 3 is 35.) (a) CH 3 MgBr + H ?...
-
1. (30 points) The Donner Family Restaurant produces hamburgers in a competitive market. The current market price of hamburgers is $6, the firm sells 1000 hamburgers per month, and is making $0 in...
-
Write down the summary of Sommers video interview with Sheila Cochran and Michael Rosen, "The Rise and Fall of Black Milwaukee's Blue Collar Middle Class"
-
read 5 reasons why the world is running out of chips on YouTube. What are the five reasons mentioned in the video that caused the current global chip shortage? Based on the current price and stocks,...

Study smarter with the SolutionInn App


