In each of the following sets, arrange the compounds in order of increasing acidity (decreasing pK a
Question:
In each of the following sets, arrange the compounds in order of increasing acidity (decreasing pKa). Explain your choices.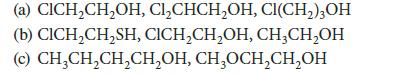
Transcribed Image Text:
(a) CICH₂CH₂OH, Cl₂CHCH₂OH, CI(CH₂)3OH (b) CICH₂CH₂SH, CICH₂CH₂OH, CH₂CH₂OH (c) CH₂CH₂CH₂CH₂OH, CH₂OCH₂CH₂OH
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 100% (1 review)
a b c The principle is that the polar effect of a chloro substituent enhances acidity the effe...View the full answer

Answered By

Utsab mitra
I have the expertise to deliver these subjects to college and higher-level students. The services would involve only solving assignments, homework help, and others.
I have experience in delivering these subjects for the last 6 years on a freelancing basis in different companies around the globe. I am CMA certified and CGMA UK. I have professional experience of 18 years in the industry involved in the manufacturing company and IT implementation experience of over 12 years.
I have delivered this help to students effortlessly, which is essential to give the students a good grade in their studies.
3.50+
2+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
Arrange the compounds in each of the following sets in order of decreasing pKa, highest first. Explain your reasoning. (a) CLCH2CH2SH CH3CH2OH CH3CH2SH (b) CH,CH,OH (CH3),N-CH-CH,OH (CH3)N OH
-
Arrange the compounds in each of the following groups in order of increasing solubility in water, and briefly explain your answers: a. 1-octanol; ethanol; ethyl chloride b. HOCH2(CHOH)3CH2OH;...
-
Arrange the bonds in each of the following sets in order of increasing polarity: (a) C-F, O-F, Be-F (b) O-Cl, S-Br, C-P (c) C-S, B-F, N-O
-
Five Smithtown High School students are saving up to buy their first cars. They all have after-school jobs, and their weekly salaries are listed in the table. Emily ..........................$110 Sam...
-
A successful businessman in the community has contacted the Moose County Board of Commissioners about donating income producing securities to the County to support a particular activity. Under the...
-
Westley Fong, manager of The Lucky 88 Motel, has a contract with Appraisers Associates to appraise his 150-room motel, which is located in beautiful downtown Wahiawa. The consultant on the job has...
-
Consider the following cash flow profile and assume MARR is 10 percent/year and the finance rate is 4 percent/ year. a. Determine the MIRR for this project. b. Is this project economically...
-
Thom DeBusk, an architect, is considering buying, restoring, and reselling a home in the Draper-Preston historic district of Blacksburg, VA. The cost of the home is $240,000 and Thom believes it can...
-
2. Two 2.5 kg masses are connected through a string as shown below. The magnitude of the force of friction on the ramp is 4.85 N. If the ramp is 5m long and mass 2 is already half way up the ramp,...
-
Use oxidation numbers to verify that the transformation in Eq. 10.39 (also shown below) is an oxidation. HC-CH-OH ethanol O || HC-C-OH acetic acid (10.39)
-
Name the following compounds. (a) Ca(OCH 3 ) 2 (b) CuSCH 2 CH 3
-
For the following exercises, find the reference angle, the quadrant of the terminal side, and the sine and cosine of each angle. If the angle is not one of the angles on the unit circle, use a...
-
Explain the role of trust in team management.
-
Prefect on the relevance of business ethics.
-
Comment on empowerment.
-
Comment on the conditions appropriate to (a) unstructured decisions and (b) structured decisions.
-
In what way does compliance differ from internalization when the focus is on sources of attitude change?
-
Benchmark Industries manufactures large workbenches for industrial use. Wayne Garrett, Benchmarks vice president for marketing, has concluded from his market analysis that sales are dwindling for the...
-
Answer the following two independent questions. a. MM Corporation is considering several proposed investments for the coming budget year. MM produces electrical apparatus for industrial complexes....
-
When optically active (R)-2-rnethylcyclohexanone is treated with either aqueous base or acid, racemization occurs. Explain.
-
Would you expect optically active (S)-3-methylcyclohexanonc to be race-mized on acid or base treatment in the same way as 2-methylcyclohexanonc. Explain.
-
When an optically active carboxylic acid such as (R)-2-phcnylpropanoic acid is brominated under HellVolhardZelinskii conditions, is the product optically active or racemic? Explain.
-
. Write the original IPv6 for the following simplified address: a. ABC:123:0:0:C0:: b. 20:CDE::1:0:3F
-
d) Find the size of the delay of the code segment below (in microconds) assuming 100 MHz internal clock frequency: Note that nop is a icycle command that just wastes the processor's time DELAY movlw...
-
a) How much memory in Tbytes can be accessed using 4 hex symbols for memory addressing assuming 8 bits (or I byte) capacity for each memory location. (Spoints) b) Please show the first 10 locations...

Study smarter with the SolutionInn App


