Name the following compound using IUPAC substitutive nomenclature. HC=CCH,CH,CH, I CH,CH,CH,CH,CH,
Question:
Name the following compound using IUPAC substitutive nomenclature.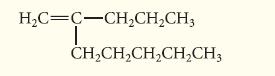
Transcribed Image Text:
HỌC=C−CH,CH,CH, I CH,CH,CH,CH,CH,
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 100% (1 review)
The principal chain is the longest continuous carbon chain cont...View the full answer

Answered By

PALASH JHANWAR
I am a Chartered Accountant with AIR 45 in CA - IPCC. I am a Merit Holder ( B.Com ). The following is my educational details.
PLEASE ACCESS MY RESUME FROM THE FOLLOWING LINK: https://drive.google.com/file/d/1hYR1uch-ff6MRC_cDB07K6VqY9kQ3SFL/view?usp=sharing
3.80+
3+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
Name the following molecules according to the IUPAC nomenclature system. (a) (b) (c) (d) (e) (f) CH32 Cl CH3 Br
-
Give an IUPAC substitutive name for each of the following alcohols: (a) (b) (c) (d) (e) (f) OH HO HO
-
Give an IUPAC substitutive name for each of the following compounds. Include the E,Z designations where appropriate. (a) (b) (c) CHi CH3 CH,CH,CH,CH C CH C-C H,C CH3
-
Can public works increase equilibrium wages?
-
Caldwell was shopping in a K-Mart store, carrying a large purse. A security guard observed her look at various small items such as stain, hinges, and antenna wire. On occasion she bent down out of...
-
The Ace Company sells a single product, at a budgeted selling price per unit of $20. Budgeted fixed manufacturing costs for the coming period are $10,000, while budgeted fixed marketing expenses for...
-
Reconsider Problem 56. Determine which oven should be purchased based on an incremental annual worth analysis. Data from problem 56 Octavia Bakery is planning to purchase one of two ovens. The...
-
Empire Company is a manufacturer of smart phones. Its controller resigned in October 2017. An inexperienced assistant accountant has prepared the following income statement for the month of October...
-
4. Ethan bought new car worth $60 000. After 5 years, the car was worth $35 429.40. Calculate the depreciation rate of the Ethan's car. [T4] [C2]
-
Which of the following alkenes can exist as double-bond stereoisomers? Identify the stereocenters in each. (a) H,C=CHCH,CH,CH, (b) CH,CH,CH=CHCH,CH, 1-pentene 3-hexene (c) HC=CH-CH=CH-CH3 (d)...
-
Arrange the labeled bonds in the following molecule in order of increasing length, shortest first. Explain your reasoning. Ha H H C=C H CH,CH,
-
In Problems 2534, find the exact value of each expression. tan 20 + tan 25 1 tan 20tan 25
-
7c. (5 pts) Assume that f is some function that takes one float argument and returns a float result (one example is def f(x) : return X ***2). Explain what the expression below computes using the...
-
How many kilograms of water must evaporate from a 49.2 kg woman to lower her body temperature by 0.708C? The latent heat of vaporization for water at 37C is 580 kcal/kg and the specific heat of the...
-
Bedrock Incorporated is owned equally by Barney and his wife Betty, each of whom holds 550 shares in the company. Betty wants to reduce her ownership in the company, and it was decided that the...
-
Java Web Homework requirements: Create any website using servlet, jsp technologies, as well as advanced frameworks such as spring, spring boot sturts 2. The website must include a homepage, login...
-
Burlington Paper Goods is considering purchasing a new delivery truck. Burlington uses the average rate of return method to evaluate capital asset decisions. If the initial cost of the truck is...
-
Explain the law of supply. Why does the supply curve slope upward? How is the market supply curve derived from the supply curves of individual producers?
-
Jax Incorporated reports the following data for its only product. The company had no beginning finished goods inventory and it uses absorption costing. $ 57.30 per unit $ 10.30 per unit $ 7.80 per...
-
A1, 2-cis disubstituted cyclohexane, such as cis-1, 2-dichlorocyclohexane, must have one group axial and one group equatorial. Explain.
-
A 1, 2-trans disubstituted cyclohexane must have either both groups axial or both groups equatorial. Explain.
-
Why is a 1, 3-cis disubstituted cyclohexane more stable than its transisomer?
-
1. (4) For the QRS peak values listed below: a. |=-10 mm, Il=+5 mm, III=15 (1) Calculate the mean cardiac vector angles according to convention given below: b(2) 1+20 mm, II=?, III=-15 mm(use...
-
Discuss the role of change management in facilitating successful organizational transitions and improvements.
-
1) What are the challenges in achieving EDM? 2) What are the business drivers for Cloud Computing? 3) What are some of the Big Data guidelines for Enterprise Decision Makers? 4) Why is EDM a...

Study smarter with the SolutionInn App


