Outline a mechanism for the reaction shown in Fig. P24.58, which is an example of the Maillard
Question:
Outline a mechanism for the reaction shown in Fig. P24.58, which is an example of the Maillard reaction followed by the Amadori rearrangement.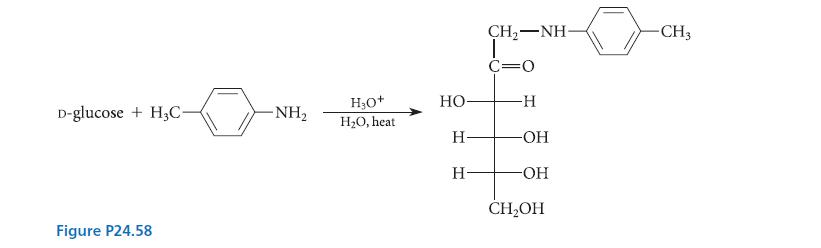
Transcribed Image Text:
D-glucose+H3C- Figure P24.58 -NH₂ H₂O + H₂O, heat HO- H- H- CH, NH T C=O -H -OH -OH CH₂OH -CH3
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 100% (QA)
The reaction of an amine with an aldose gives a carbinolamine which dehydrates to an iminium ion A T...View the full answer

Answered By

Usman Nasir
I did Master of Commerce in year 2009 and completed ACCA (Association of Chartered Certified Accountants) in year 2013. I have 10 years of practical experience inclusive of teaching and industry. Currently i am working in a multinational company as finance manager and serving as part time teacher in a university. I have been doing tutoring via many sites. I am very strong at solving numerical / theoretical scenario-based questions.
4.60+
16+ Reviews
28+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
The following is an example of a reaction sequence developed by Derin C. D'Amico and Michael E. Jung (UCLA) that results in enantiospecific formation of two new chirality centers and a carbon-carbon...
-
Write a mechanism for the reaction shown in eq. 10.35. room C-OCH, +HCl (10.35) temp benzoyl chloride methyl benzoate
-
(a) Give a curved-arrow mechanism for the reaction shown in Fig. P11.70. Be sure your mechanism indicates the role of the weak acid ammonium chloride. (b) Why does the reaction of an aziridine...
-
Use the same information as in question 4, except that Hughes has a debit balance of $5,000 in its Allowance for Doubtful Accounts before any adjustments are made at the end of the year. In this...
-
Suppose that two billion years ago 10% of the mass of the earth were 14 C. Approximately what percent of the mass of the earth today would be 14 C, neglecting formation of 14 C in the atmosphere?
-
Jasmine Scents has been given two competing offers for short-term financing. Both offers are for borrowing $15,000 for 1 year. The first offer is a discount loan at 8%; the second offer is for...
-
Prove that, for any \(\beta>-1\), any bounded Borel function \(h\), and any bounded Borel function \(\varphi\) valued in \(]-1, \infty[\), the processes \[\begin{aligned} & \exp \left[\ln (1+\beta)...
-
Compute the early, late, and slack times for the activities in the network that follows, assuming a time-constrained network. Which activities are critical? What is the time-constrained project...
-
Extracts from the financial statements for Bridgestone Plc for the year ended 31 December are as follows: Income Statement for the year ended 31 December 2020 Operating profit Interest expense Profit...
-
Maltose is a disaccharide obtained from the hydrolysis of starch. Maltose can be hydrolyzed to two equivalents of glucose and can be oxidized to an acid, maltobionic acid, with bromine water....
-
L-Rhamnose is a 6-deoxyaldose with the following structure. When a methyl glycoside of l-rhamnose, methyl a-l-rhamnopyranoside, was treated with periodic acid, compound A, C 6 H 12 O 5 , was obtained...
-
Petcetera had a robbery on the weekend in which a large amount of inventory was taken. The loss is totally covered by insurance. A physical inventory count determined that the cost of the remaining...
-
Was your organization prepared, actively and actionably, to deal with the Covid-19 crisis? What should or could have been done to better prepare?
-
What is the concept of product stewardship, and how can it be achieved?
-
What do you think about the authors argument about the generational differences in consumer expectations of business?
-
Do you think that the characteristics, as described throughout the chapter, perpetuate stereotypes of women, why or why not?
-
Do you use mindfulness in your routine practices of leadership and management?
-
The Shell Oil Company, which began about 1912, had been for decades a household name as a quality oil company in the United States. However, by the late 1970s much of its prestige as a premiere...
-
Use translations to graph f. f(x) = x-/2 +1
-
Give the product formed when the following alcohols is oxidized by dilute HNO3. HOCH2CH 2CH2OH
-
Assuming you knew the properties of the compound obtained in Problem 24.23, including its optical rotation, show how you could use periodate oxidation to distinguish methyl -D- galactopyranoside from...
-
Give the products expectedly when n-ribose (or other compolrnd indicated) reacts with each of the following reagents. (a) Dilute HNO-, (b) -CN,H2O (c) Product of part (b) + H2/Pd/BaSO4 + H2O +/H2O...
-
Ratio analysis compares line-item data from a company's financial statements to reveal insights regarding profitability, liquidity, operational efficiency, and solvency. UBER TECHNOLOGIES, INC....
-
Given data below answer the question. Job Letter Description Normal Time (days) Immediate Predecessor(s) A Start 0.0 B Design 8.0 A C Order special accessories 0.1 D Build frame 1.0 E Build doors 1.0...
-
You own a boba smoothie store. Your line up of smoothies is similar with those offered at Jamba Juice or Juice It Up. The difference is, you have different kinds of boba (small boba, big boba, jelly,...

Study smarter with the SolutionInn App


