Outline a synthesis of (cyclohexylmethyl)methylamine from cyclohexanecarboxylic acid. 010 OH CH-NHCH3 cyclohexanecarboxylic acid (cyclohexylmethyl)methylamine
Question:
Outline a synthesis of (cyclohexylmethyl)methylamine from cyclohexanecarboxylic acid.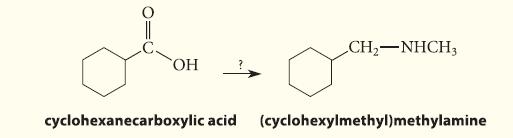
Transcribed Image Text:
010 OH CH₂-NHCH3 cyclohexanecarboxylic acid (cyclohexylmethyl)methylamine
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 0% (1 review)
Any carboxylic acid derivative used to prepare the amine must contain ...View the full answer

Answered By

JAPHETH KOGEI
Hi there. I'm here to assist you to score the highest marks on your assignments and homework. My areas of specialisation are:
Auditing, Financial Accounting, Macroeconomics, Monetary-economics, Business-administration, Advanced-accounting, Corporate Finance, Professional-accounting-ethics, Corporate governance, Financial-risk-analysis, Financial-budgeting, Corporate-social-responsibility, Statistics, Business management, logic, Critical thinking,
So, I look forward to helping you solve your academic problem.
I enjoy teaching and tutoring university and high school students. During my free time, I also read books on motivation, leadership, comedy, emotional intelligence, critical thinking, nature, human nature, innovation, persuasion, performance, negotiations, goals, power, time management, wealth, debates, sales, and finance. Additionally, I am a panellist on an FM radio program on Sunday mornings where we discuss current affairs.
I travel three times a year either to the USA, Europe and around Africa.
As a university student in the USA, I enjoyed interacting with people from different cultures and ethnic groups. Together with friends, we travelled widely in the USA and in Europe (UK, France, Denmark, Germany, Turkey, etc).
So, I look forward to tutoring you. I believe that it will be exciting to meet them.
3.00+
2+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
Outline a synthesis of each of the following compounds from the indicated starting materials and any other reagents. (a) 1-cyclohexyl-2-methyl-2-prupanol from bromocyclohexane (b) PhNHCH2CH2CH(CH3)2...
-
Outline a synthesis of each of the following compounds from isobutyric acid (2-methylpropanoic acid) and any other necessary reagents. (a) (b) 0 CH) CHC OCH, CCHCH isobutyrophenone
-
Outline a synthesis of phenylethyne from each of the following: (a) (b) (c) (d) Br Br Br
-
Assignment: Based on your reading and analysis of the case study above, address the following items in a detailed essay response of approximately 600 words. Each number below should be addressed...
-
Consider the following incomplete table of merchandisers profit data: Requirement 1. Calculate the missing table values to complete thetable. Cost of Goods Sold Net Sales Gross Profit Sales Sales...
-
Consider the balance sheet for the DI listed below: The DI is expecting a $ 15 million net deposit drain. Show the DI's balance sheet under these two conditions: a. The DI purchases liabilities to...
-
Redesign the fractionator of Example 6.8 for a reflux ratio that is twice the minimum. Determine the diameter of the tower, the height of packing in the stripping and rectifying sections, and the...
-
On January 1, 2008, Lennon Industries had stock outstanding as follows. 6% Cumulative preferred stock, $100 par value, issued and outstanding 10,000 shares ....$1,000,000 Common stock, $10 par value,...
-
Using the list method or the switch method, find the inverse function of: f(x)=6x+1-13 f1(x) =
-
A water-insoluble hydrocarbon A decolorizes a solution of Br 2 in CH 2 Cl 2 . The base peak in the EI mass spectrum of A occurs at m/z = 67. The proton NMR of A is complex, but integration shows that...
-
Isopentenyl pyrophosphate, the starting material for isoprenoid and steroid biosynthesis (Sec. 17.6B), is formed biosynthetically by the decarboxylation reaction of...
-
In an experiment to determine the calorific value of octane \(\left(\mathrm{C}_{8} \mathrm{H}_{18} ight)\) with a bomb calorimeter the mass of octane was \(5.42195 \times 10^{-4} \mathrm{~kg}\), the...
-
explain why monetary autonomy is impossible on its own with a fixed exchange rate using IS/LM or the trilemma, and why monetary policy must accompany fiscal policy with a fixed exchange rate
-
what happens to the demand and the demand curve for beef if more people decide to become vegans ?
-
What happens if a toaster company makes 10,000 toasters to sell for $80 and they only sell 7,000? What would they do with the other 3,000 toasters?
-
Calculate the impulse experienced when a 68 kg person lands on firm ground after jumping from a height of 2.5 m Express your answer using two significant figures. J = Submit Part B Favg Submit Part...
-
suppose you are at a bundle, such that MRS>MRT, how should you change your consumption so as to maximize utility subject to your budget constraint?
-
What does the term sales mix mean? How is a weighted average unit contribution margin computed?
-
During 2012, Cheng Book Store paid $483,000 for land and built a store in Georgetown. Prior to construction, the city of Georgetown charged Cheng $1,300 for a building permit, which Cheng paid. Cheng...
-
The heat of hydrogenation of allene is 298 kJ mol-1, whereas that of propyne is 290 kJ mol-1. (a) Which compound is more stable? (b) Treating allene with a strong base causes it to isomerize to...
-
Although both 1-bromobutane and 4-bromo-1-butene are primary halides, the latter undergoes elimination more rapidly. How can this behavior be explained?
-
Complete the molecular orbital description for the ground state of cyclopentadiene shown at right. Shade the appropriate lobes to indicate phase signs in each molecular orbital according to...
-
Four people are attempting to move a stage platform across the floor. If they exert the horizontal forces shown, determine (a) the equivalent force-couple system at O and (b) the points on the x- and...
-
When calculating Sell-thru, it is best to grouIn the weeks leading up to Easter, the store did a special order of 600 12-lb spiral-cut hams. The hams were priced at $3.50/lb. 85% of the hams sold for...
-
Problem 3 [5 points] (Laplace transform) A linear time-invariant (LTI) system has input x(t), impulse response h(t), and output y(t). Assume that the input is given by: x(t) = eu(t) where u(t) is the...

Study smarter with the SolutionInn App


