Provide reaction mechanisms for the following equations. (a) Eq. 24.12a (b) Eq. 24.12b H3C-CH-CH2CH2CH2-CH=0 T OH H3C-CH-CH2CH2-CH=0
Question:
Provide reaction mechanisms for the following equations.
(a) Eq. 24.12a
(b) Eq. 24.12b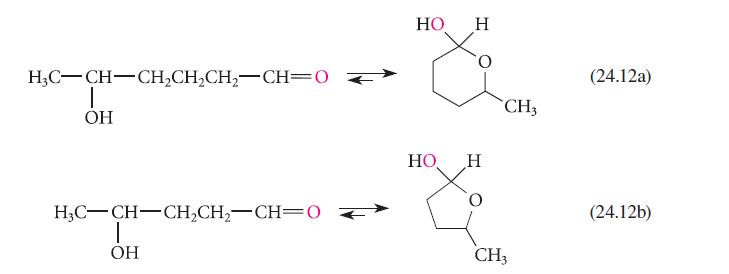
Transcribed Image Text:
H3C-CH-CH2CH2CH2-CH=0 T OH H3C-CH-CH2CH2-CH=0 T OH HO HO Н H 0 CH3 CH3 (24.12a) (24.12b)
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 66% (3 reviews)
a HC CH CHCHCH ...View the full answer

Answered By

Ankur Gupta
I have a degree in finance from a well-renowned university and I have been working in the financial industry for over 10 years now. I have a lot of experience in financial management, and I have been teaching financial management courses at the university level for the past 5 years. I am extremely passionate about helping students learn and understand financial management, and I firmly believe that I have the necessary skills and knowledge to effectively tutor students in this subject.
4.80+
2+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
Provide reaction mechanisms for the following equations. (a) Eq. 23.17 (b) Eq. 23.18 (c) Suggest a reason why the reaction in Eq. 23.18 stops after two additions, and a third doesnt occur in high...
-
uestion text Pepper Company owned 60,000 of Salt Companys 100,000 outstanding shares On January 2, 2018, Salt purchased 20,000 of its outstanding shares from the NCI for $70,000 Pepper purchased its...
-
The Blacks moved from Maine to Nevada. As a result, they sold their house in Maine on January 4, 2018. They originally paid $76,000 for the home on July 3, 1993, but managed to sell it for $604,000....
-
Write a method remove() that takes a linked-list Node and a string key as its arguments and removes every node in the list whose item field is equal to key.
-
Two protons approach each other head on at 0.5c relative to reference frame S. (a) Calculate the total kinetic energy of the two protons as seen in frame S. (b) Calculate the total kinetic energy of...
-
Sam and Suzy Sizeman need to prepare a cash budget for the last quarter of 2013 to make sure they can cover their expenditures during the period. Sam and Suzy have been preparing budgets for the past...
-
A 55 g soapstone cubea whisky stoneis used to chill a glass of whisky. Soapstone has a density of 3000 kg/m 3 , whisky a density of 940 kg/m 3 . What is the approximate normal force of the bottom of...
-
Entries for Lower-of-Cost-or-Market'Direct and Allowance Malone Company determined its ending inventory at cost and at lower-of-cost-or-market at December 31, 2009, December 31, 2010, and December...
-
3. Draw a full 3-ary tree with 20 vertices. List the root, all internal vertices, leaves, children of one level 1 vertex, parent of one level 2 vertex, descendants of one non-leaf vertex, ancestors...
-
Consider the -D-pyranose forms of glucose and talose (Fig. 24.3). Suggest one reason why talose contains a smaller fraction of -pyranose form than glucose. - - H CH=0 -OH HO OH OH -OH CHOH H CH-O...
-
Which pair of the following aldoses are epimers and which pair are enantiomers? H HO H CH=0 -OH A - -OH -CHOH 0=CH- - OH H B - -H -OH -CHOH H - OH -CH=0 -OH - - CHOH
-
For each of the following situations, prepare the adjusting entry for the month ended May 3l and indicate the effect each adjustment would have on net income: A. The May telephone bill for Scheele...
-
What are the arguments for and against rules?
-
According to the Federal Reserve Act of 1913 (Section 13.3), In unusual and exigent circumstances, the Board of Governors of the Federal Reserve System, [] may authorize any Federal Reserve bank,...
-
What is frictional unemployment? Why can it be beneficial for workers, firms, and the economy?
-
Why do firms hold inventories, and why is their inventory investment a matter of interest to macroeconomists?
-
What are the key ideas of the real business cycle model? How does it explain business cycle fluctuations?
-
According to Student Monitor, a New Jersey research firm, the average cumulated college student loan debt for a graduating senior is $25,760. Assume that the standard deviation of such student loan...
-
Borrowing costs should be recognised as an expense and charged to the profit and loss account of the period in which they are incurred : A. If the borrowing costs relate to qualifying asset B. If the...
-
For each of the following structures, 1. Draw a Lewis structure; fill in any nonbonding electrons. 2. Calculate the formal charge on each atom other than hydrogen. All are electrically neutral except...
-
1. From what you remember of electronegativities, show the direction of the dipole moments of the following bonds. 2. In each case, predict whether the dipole moment is relatively large...
-
Determine whether the following pairs of structures are actually different compounds or simply resonance forms of the same compounds. (a) (b) (c) (d) (e) (f) (g) (i) (j) and O- and and O- CH C H and...
-
Directions: Be sure to save an electronic copy of your answer before submitting it to Ashworth College for grading. Unless otherwise stated, answer in complete sentences, and be sure to use correct...
-
You plan to retire in 2 5 years and you want to receive $ 7 , 0 0 0 per month for the remainder of your life ( 3 5 years in retirement ) . The first payment is received the day you retire. Assuming a...
-
PLANNING BUSINESS MESSAGES. Topic 1: Following the 3-x-3 writing process Topic 2: Analyzing the Purpose and anticipating the audience Topic 3: Improving the Tone and Clarity of a Message. Describe...

Study smarter with the SolutionInn App


