Which pair of the following aldoses are epimers and which pair are enantiomers? H HO H
Question:
Which pair of the following aldoses are epimers and which pair are enantiomers?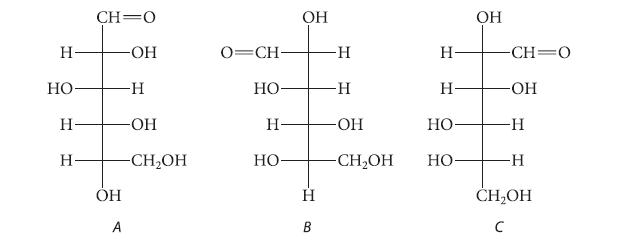
Transcribed Image Text:
H HO H Н CH=0 -OH ОН A -Н -OH -CH₂OH 0=CH- НО н- НО OH H B -Н -H -OH -CH₂OH H Н- Но НО OH -CH=0 -OH -Н -Н CH₂OH с
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 0% (1 review)
Use cyclic permutations to put all of the aldoses in the standard Fischer p...View the full answer

Answered By

Munibah Munir
I've done MS specialization in finance’s have command on accounting and financial management. Forecasting and Financial Statement Analysis is basic field of my specialization. On many firms I have done real base projects in financial management field special forecasting. I have served more than 500 Clients for more than 800 business projects, and I have got a very high repute in providing highly professional and quality services.I have capability of performing extra-ordinarily well in limited time and at reasonable fee. My clients are guaranteed full satisfaction and I make things easy for them. I am capable of handling complex issues in the mentioned areas and never let my clients down.
4.60+
467+ Reviews
648+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
For each pair of the following compounds identify which compound would react more rapidly in an E1 reaction. a. b. CI .CI CI CI
-
For each pair of the following compounds identify which compound would react more rapidly in an E2 reaction: a. b. CI .CI Br Br
-
Draw and name the pair of epimers formed when the following aldopentoses undergo a Kiliani Fischer chain-lengthening process: (a) (b) (c) . %3D - . - - - CH- D-Ribose . %3D - - - H,OH D-Xylose
-
Suppose that \(\mathrm{x}\) is a linked-list Node. What is the effect of the following code fragment? \(t\). next \(=x\). next; \(x \cdot\) next \(=t ;\)
-
The sun radiates energy at the rate of about 4 10 26 W. Assume that this energy is produced by a reaction whose net result is the fusion of 4 H nuclei to form 1 He nucleus, with the release of 25...
-
The actual sales and purchases for Xenocore, Inc., for September and October 2012, along with its forecast sales and purchases for the period November 2012 through April 2013, follow. The firm makes...
-
You are looking for a deep spot to take a swim in a local river. The width of the river is about the same all along its length, but there are places where the water flows quickly and places where it...
-
Each fall the Draperton Parks and Recreation Department holds a series of tryouts for its boys' and girls' youth basketball leagues. All leagues are formed by age group, each usually encompassing a...
-
6) Draw a timing diagram for x1, x2, x3, and f, for the circuit shown in the figure. x2 x3 x D D f
-
Provide reaction mechanisms for the following equations. (a) Eq. 24.12a (b) Eq. 24.12b H3C-CH-CH2CH2CH2-CH=0 T OH H3C-CH-CH2CH2-CH=0 T OH HO HO H 0 CH3 CH3 (24.12a) (24.12b)
-
Which of the following are Fischer projections of a meso compound? - - - CH-0 - -OH - CHOH A - - CHOH - -OH - CHOH B - - - -CHOH - - CHOH - - HOCH -CHOH - -
-
Using the information provided in Part 1 of Problem 1B journalize each of the transactions assuming a periodic inventory system. In Problem 1B Part 1 Prepare General Journal entries to record the...
-
What has been the general experience of countries that have adopted inflation targeting?
-
What are the arguments for and against central bank independence?
-
Most legal systems assume that it is better not to incarcerate a guilty individual than to incarcerate an innocent person (i.e., if you are making a mistake, at least choose the lesser of the two)....
-
Why is the quantity of labor demanded inversely related to the real wage rate?
-
How do new Keynesian ideas about expectations affect the IS and aggregate demand curves?
-
Data accumulated by the National Climatic Data Center shows that the average wind speed in miles per hour for St. Louis,Missouri, is 9.7. Suppose wind speed measurements are normally distributed for...
-
Could the owner of a business prepare a statement of financial position on 9 December or 23 June or today?
-
Draw the important resonance forms to show the delocalization of charges in the following ions. (a) (b) (c) (d) (e) (f) (g) (h) (i) (j) CH-C CH H-C-CH CHCH CH2 NH CHCH- CH CH CH-CH 3 CH3 CH CH CH CH...
-
All of the following compounds can react as acids. Without using a table of acidities, rank them in order of increasing acidity. Explain your ranking. (a) CH3CH2SO3H (b) CH3CH2OH (c) CH3CH2COOH (d)...
-
The following compound can become protonated on any of the three nitrogen atoms. One of these nitrogens is much more basic than the others, however. (a) Draw the important resonance forms of the...
-
A project with an initial cost of $30,000 promises to return $5,000; $10,000; $11,000; $13,000; and $17,000 in years 1 through 5 respectively, and nothing after that. a. Draw the cash flow diagram....
-
What actions could JP Chase & Morgan have used to lessen the impact of the 2 0 1 3 financial loss? Explain
-
SUCCEEDING IN THE SOCIAL AND MOBILE WORKPLACE. Topic 1: Strong communication skills are key to success. Topic 2: Developing listening skills. Topic 3: Becoming interculturally proficient. describe...

Study smarter with the SolutionInn App


