Provide two different reaction sequences that could be used to synthesize 4-methoxy-3-methylbiphenyl. Both sequences, however, should start
Question:
Provide two different reaction sequences that could be used to synthesize 4-methoxy-3´-methylbiphenyl. Both sequences, however, should start with both p-bromoanisole and m-bromotoluene.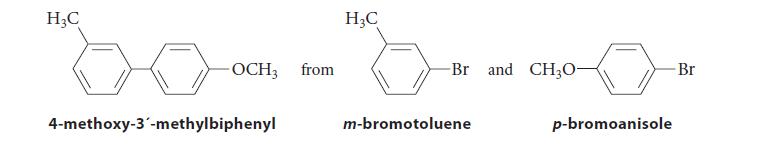
Transcribed Image Text:
H₂C -OCH3 from 4-methoxy-3'-methylbiphenyl H₂C -Br and CH3O- m-bromotoluene p-bromoanisole Br
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 50% (2 reviews)
The starting materials for a Suzuki coupling can be determined by mentally splitting the arylar...View the full answer

Answered By

Mugdha Sisodiya
My self Mugdha Sisodiya from Chhattisgarh India. I have completed my Bachelors degree in 2015 and My Master in Commerce degree in 2016. I am having expertise in Management, Cost and Finance Accounts. Further I have completed my Chartered Accountant and working as a Professional.
Since 2012 I am providing home tutions.
3.30+
2+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
Write a literature review for your study. See below for an example of a literature review. Your literature review should provide both analysis and synthesis of previous studies as related to the...
-
In 2015, John decided to start up his own brewery, Tasmania Microbrewery Inc (TMI). His family supported his decision and joined him in investing in the business. TMI began operations on January 1,...
-
In 2009, David Stott quit his job at a large beer company to start his own brewery, Arizona Microbrewery, Inc. (AMI). His family supported his decision and invested in the business along with David....
-
Use a software package such as Matlab or Mathematica to program the example described in section 7.3. (a) Assume the environmental regulator ignores the impact on the labor market and sets an...
-
A spaceship of proper length L = 400 m moves past a transmitting station at a speed of 0.76c. At the instant that the nose of the ship passes the transmitter, clocks at the transmitter and in the...
-
Kelsey Drums, Inc., is a well-established supplier of fine percussion instruments to orchestras all over the United States. The companys class A common stock has paid a dividend of $5.00 per share...
-
For the given set of periodic processes, all of which share the same deadline of 12: a. Schedule the processes for the given arrival times using standard ratemonotonic scheduling (no data...
-
Beta Industries has net income of $2,000,000, and it has 1,000,000 shares of common stock outstanding. The companys stock currently trades at $32 a share Beta is considering a plan in which it will...
-
Marwick's Pianos, Incorporated, purchases pianos from a manufacturer for an average cost of $1,505 per unit and then sells them to retail customers for an average price of $2,600 each. The company's...
-
Draw a curved-arrow mechanism for the last (acid-catalyzed hydrolysis) step of Eq. 18.50. MgBr OCH3 + B(OCH3)3 B(OCH3)3 MgBr OCH3 H0 HO B(OH)2 OCH3 + 3 CH3OH 2+ + Mg+ + Br (18.50)
-
What product is expected when cyclopentene reacts with iodobenzene in the presence of triethylamine and a Pd(0) catalyst?
-
Solve Prob. 598 for the maximum shear stress within regions AC and BC, and the angle of twist Ï of end B relative to C. A. 20 N-m 50 N-m 30 N-m 50 mm mm 1.5 m
-
The amount of gain (loss) taken to equity in respect of the put option for the year ended 31 December 20x5 was: (a) $0 (b) Gain of $38,000 (c) Gain of $12,000 (d) Gain of $50,000 (e) None of the...
-
What should be the total cost of the equipment as at 1 February 20x5 under IFRS 9? (a) $12,800,000 (b) $12,500,000 (c) $11,500,000 (d) $12,300,000 On 1 October 20x4, SingCo committed to purchase...
-
What is the gain/(loss) to be recognized in the income statement for 20x4 under IFRS 9? (a) $800,000 (b) 0 (c) $(100,000) (d) $(200,000) (e) $100,000 On 1 October 20x4, SingCo committed to purchase...
-
On 30 September 20x5, Singco entered into a non-cancellable contract to purchase inventory for 100,000 euros to be delivered on 31 January 20x6 with payment due on 31 March 20x6. Singco was concerned...
-
Alfalfa Companys functional currency is the dollar. On 30 June 20x1, it entered into a forward exchange contract to purchase FC 100,000 at the forward rate of $1.077 for delivery on 30 June 20x2. The...
-
According to Padgett Business Services, 20% of all smallbusiness owners say the most important advice for starting a business is to prepare for long hours and hard work. Twenty-five percent say the...
-
Read the following description and Write a response of it. The discretion of public administrators can be decreased, but not altogether eliminated. Officials will use their discretion in any given...
-
(a) Write the propagation steps leading to the formation of dichloromethane (CH2Cl2) from chloromethane. (b) Explain why free-radical halogenations usually gives mixtures of products. (c) How could...
-
Draw resonance forms to show how the BHA radical is stabilized by delocalization of the radical electron over other atoms in the molecule.
-
The triphenylmethyl cation is so stable that some of its salts can be stored for months. Explain why this cation is so stable. triphenylmethyl cation
-
An investment broker that Ava trusts recommended that she purchase a $ 5 0 , 0 0 0 , 1 5 - year municipal bond that generates a dividend of 6 % per year payable quarterly. She will pay a discounted...
-
Rael Corp had the following balances in its accounts for gross accounts receivables and the allowance for doubtful accounts at the beginning of the year. Accounts Receivable Allowance for...
-
A force of 14.0 N directed down a ramp inclined at 20 pushes on a 19.5 kg box; the box is at the point where it is just about to move. Determine the coefficient of static friction between the box and...

Study smarter with the SolutionInn App


