Rank each of the following compounds in order of increasing S N 1 solvolysis reactivity in ethanol,
Question:
Rank each of the following compounds in order of increasing SN1 solvolysis reactivity in ethanol, and explain your choices by drawing suitable structures.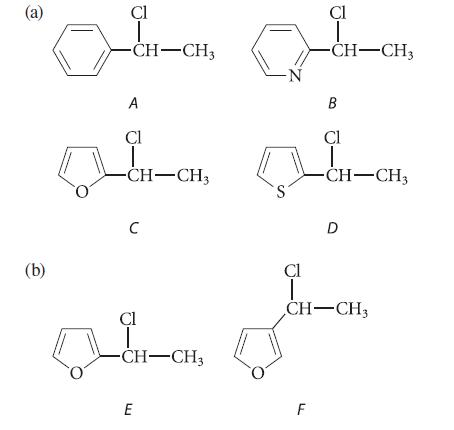
Transcribed Image Text:
(a) (b) Cl T -CH-CH3 A Cl CH-CH3 C Cl -CH-CH₂ E -N S Cl T -CH-CH3 F B Cl T -CH-CH3 D Cl T CH-CH3
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 100% (1 review)
a The order ...View the full answer

Answered By

John Aketch
I have a 10 years tutoring experience and I have helped thousands of students to accomplish their educational endeavors globally. What interests me most is when I see my students being succeeding in their classwork. I am confident that I will bring a great change to thins organization if granted the opportunity. Thanks
5.00+
8+ Reviews
18+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
Rank the following compounds in order of increasing reactivity (least reactive first) in an SN1 solvolysis reaction in aqueous acetone. Explain your answers. (The structure of tert-cumyl chloride is...
-
Rank the following compounds in order of increasing reactivity toward nitration with HNO3 and explain your choices: thiophene, benzene, 3-methylthiophene, and Z-methvlfuran.
-
Rank the following compounds in order of increasing reactivity in bromination. In each case, indicate whether the principal monobromination products will be the ortho and para isomers or the meta...
-
Use Lagrange multipliers to find the maximum area of a rectangle inscribed in the ellipse (Figure 15): (-x,y) (x, y) 2 a2 b2 + J2 = 1 (x, y) (x, y) X
-
Institutionally Related Foundations. Compass State University Foundation (CSUF) was incorporated as a not-for-profit organization to support a public university in its fund-raising efforts and the...
-
Barrymore Costume Company, located in New York City, sews costumes for plays and musicals. Barrymore considers itself primarily a service firm, as it never produces costumes without a pre-existing...
-
The chemical energy of gasoline is \(46 \mathrm{MJ} / \mathrm{kg}\). If gasoline's mass could be completely converted into energy, what mass of gasoline would be needed to equal the chemical energy...
-
Martin Chicken Corporation processes and packages chicken for grocery stores. It purchases chickens from farmers and processes them into two different products: chicken drumsticks and chicken steak....
-
There are many items that we have talked about in this class. Often, we discuss terms here as they affect manufacturing companies. However, we often do not get a chance to discuss how a merchandising...
-
Would you expect Chargaffs first parity rule to apply within an individual strand of DNA? Explain.
-
Draw the structure of (a) Deoxythymidine monophosphate (dTMP); (b) GDP.
-
1. The provision of accounting information for internal users is known as a. Managerial accounting. b. Accounting. c. Financial accounting. d. Information provision. e. Accounting for planning and...
-
What other terms are used to describe deductions for adjusted gross income?
-
Is it possible to defer gain through the purchase of a new residence?
-
Explain the fruit-of-the-tree doctrine and why it was established by the courts.
-
What effect does interest on tax-exempt bonds have on the taxation of social security benefits?
-
What is a nonrecourse loan? Will a nonrecourse loan given by the seller of real estate to the buyer increase the amount the buyer has at risk? Explain.
-
Determine the SSE and the se for Problem 12.7. Use the residuals computed in Problem 12.15 (for Problem 12.7) and determine how many of them are within 1se and 2se. How do these numbers compare with...
-
Show that if A is any m n matrix, then Im A = A and AIn = A.
-
In light of the ion-pair hypothesis, how would you expect the stereochemical outcome of an Snl reaction (percent racemization and inversion) to differ from the result discussed in this section for an...
-
Predict the products expected in each of the following situations, and show the mechanism of any reaction that takes place using the curved-arrow notation. (a) 2-bromobutaneintert-butyl alcohol...
-
What substitution and elimination products (if any)might be obtained when each of the following alkyl halides is treated with sodium methoxide in methanol? (a) methyl iodide (b)...
-
Hi, please fill out the chart, so far I have these correct and the cost of goods manufactured in part one is 1,287,600. For Firm J, prepare an income statement. Note: Use cost of goods manufactured...
-
Explain how the company can improve on the following by providing two suggestions each: Inventory turnover period Trade receivables period QUESTION FOUR 4.1 (25 Marks) Complete the table for the...
-
How are the net capital gain/loss taxed for individuals? What is the tax treatment of interest on state and local government obligations? (Remember that the exempt status applies solely to state and...

Study smarter with the SolutionInn App


