Suggest a structure for a constitutional isomer of the following compound that should have greater water solubility,
Question:
Suggest a structure for a constitutional isomer of the following compound that should have greater water solubility, and explain your reasoning. The structure should not be an enol, because enols are not stable. 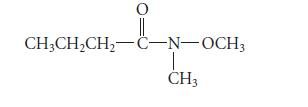
Transcribed Image Text:
O || CH3CH₂CH₂-C-N-OCH3 CH3
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 0% (2 reviews)
The compound shown in the image is Nmethylpropanamide A constitutional isomer of this compound ...View the full answer

Answered By

Nyron Beeput
I am an active educator and professional tutor with substantial experience in Biology and General Science. The past two years I have been tutoring online intensively with high school and college students. I have been teaching for four years and this experience has helped me to hone skills such as patience, dedication and flexibility. I work at the pace of my students and ensure that they understand.
My method of using real life examples that my students can relate to has helped them grasp concepts more readily. I also help students learn how to apply their knowledge and they appreciate that very much.
4.00+
1+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
The UV spectrum of 1-phenylprop-2-en-1-ol shows an intense absorption at 220 nm and a weaker absorption at 258 nm. When this compound is treated with dilute sulfuric acid, it rearranges to an isomer...
-
our class. The word count of the text that includes your "own words" should be at least 500 words (about 700 max.). The text should be typed double-spaced using a 12-point font size in Times New...
-
(a) Two amides are constitutional isomers and have the formula C 4 H 9 NO, and each contains an isopropyl group as part of its structure. Give structures for these two isomeric amides. (b) Draw the...
-
Cost-Volume-Profit (CVP) analysis can be used to determine the effect of changes in costs and volumes on a company's net profits. This project assignment assumes that you are newly hired as an...
-
Identify the questions you should ask to anticipate your audiences reaction.
-
Matilda owns a condominium on the beach in Rehoboth, Delaware. During the current year, she incurs the following expenses related to the property: Mortgage interest ...... $8,000 Property taxes...
-
Refer to the information in QS 19-16. The company sells its product for $50 per unit. Due to new regulations, the company must now incur $2 per unit of hazardous waste disposal costs and $8,500 per...
-
Gallatin Carpet Cleaning is a small, family-owned business operating out of Bozeman, Montana. For its services, the company has always charged a flat fee per hundred square feet of carpet cleaned....
-
Design Deterministic Finite Automata with for each of the following independent specifications. Design a DFA that accepts the words that have odd number of's. Design a DFA where the members of has at...
-
Vitamins can be classified as fat-soluble or watersoluble. Fat-soluble vitamins can be stored in fatty tissues, whereas water-soluble vitamins can be excreted in the urine. (a) The structures of some...
-
When salad oil is mixed with water and shaken, two layers quickly separate, the oily layer on top and the water layer on the bottom. When an egg yolk (which is rich in lecithin, a phospholipid) is...
-
Evaluate the following integrals. te dt
-
The following transactions occurred during the twelve-month accounting period ending December 31, 2016 for Hammerhead, Inc. ("Corporation"). Be sure to include in your analysis any adjusting journal...
-
A better understanding of social entrepreneurship requires a good understanding of commercial and social entrepreneurs. Although social entrepreneurs share some of the qualities of regular...
-
1. Tibo's supply chain case study(include references) Making Phones in China Tibo Inc. is an American consumer electronics company. Its most popular and flagship product is the V10 smartphone. The...
-
Retail Food Group shares suffered their biggest fall in nine years yesterday after UBS said changes to international accounting standards could cause a "material" impact on the company's debt...
-
Part 1 After reading Milton Friedman's article, take a position using your own research. Take a position "for" or "against" corporate social responsibility relative to domestic and foreign locations....
-
Explain how featherbedding and other restrictive work practices can reduce labor productivity. Why might strikes reduce the economys output less than the loss of production by the struck firms?
-
Determine the annual percentage yield for a loan that charges a monthly interest rate of 1.5% and compounds the interest monthly.
-
Provide names for thesecompounds: a) CH;CH,CH,CH,CH,CH b) CH f) CH,CCH,CCH, d) CI g) h)
-
Draw the structures for these compounds: (a) (Z)-Oct-3-en-2-one (b) 3-Ethylheptanal (c) 2, 4-Pentadienal (d) 3, 4-Dimethylbenzaldehyde (e) 1-Phenyl-1-propanone (f) 2, 2, 6, 6-Ttramythleyelohexanone
-
Explain which the most acidic hydrogen's in these compoundsare c) CH,CCH,CH, b) PHCH CCH3
-
On one graph show how supply and demand side market failures emerge. How does the market forces clear the failures for normal goods
-
If the economy is moving too slowly, which economic problem is more likely to exist-high unemployment or high inflation? In this situation, should the federal government increase taxes or decrease...
-
M MD 5600 head 5300bad area diagram 42 MD area D faria C 0 5,000 head/yr. cande in headsy 10,000 had 8. Using diagram #2, where, Mrancher (defined as MR-MC) is the expected marginal profits to the...

Study smarter with the SolutionInn App


