Tell whether the configuration of each asymmetric atom in the following compounds is R or S. (a)
Question:
Tell whether the configuration of each asymmetric atom in the following compounds is R or S.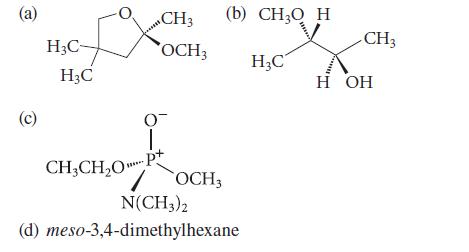
Transcribed Image Text:
(a) (c) H₂C- H3C CH3 (b) CH3Q H OCH3 0 1. CH3CH₂OP+ OCH3 N(CH3)2 (d) meso-3,4-dimethylhexane H₂C mund CH3 H OH
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 0% (1 review)
a H3C CH3 ROCH3 b CH3OH H3C S R CH3 H OH c CH3CH2O OCH3 NCH32 d An...View the full answer

Answered By

Sandip Agarwal
I have an experience of over 4 years in tutoring. I have solved more than 2100 assignments and I am comfortable with all levels of writing and referencing.
4.70+
19+ Reviews
29+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
Give the configuration of each asymmetric atom in the following compounds. (a) (b) meso-3,4- dimethlylhexane CH H,C H OH
-
Indicate whether the asymmetric atom in each of the following compounds has the R or S configuration. H . www. CH3 OH (b) NH alanine www. | CH H OH malic acid Me I ***** Pr N+ i-Pr Et CIT
-
Each of the following compounds possesses carbon atoms that are chirality centers. Locate each of these chirality centers, and identify the configuration of each one: a. b. c. d. e. f. Ephedrine A...
-
What is the future value at the end of the 20th semester of an annuity due with 20 semiannual payments of $500 each, evaluated at an annual interest rate of 10%?
-
Identify and describe the five environmental differences between governments and for-profit business enterprises as identified in the Governmental Accounting Standards Boards Why Governmental...
-
a. Why do companies generally follow a sequential strategy in moving overseas? b. What are the pluses and minuses of exporting? Of licensing? Of foreign production?
-
Several professionals are listed as being part of compliance efforts with regard to participation in the examination of potential compliance violations. Identify at least three other categories of...
-
The following transactions apply to Baker Co. for 2010, its first year of operations. 1. Issued $60,000 of common stock for cash. 2. Provided $128,000 of services on account. 3. Collected $113,200...
-
Joe is 25 and has a wage of $30,000. He is single with no dependents, and files as single. He rents and makes no contributions to charity. He contributed $800 to a Roth IRA in 2022. What is his Total...
-
(a) Draw sawhorse projections of ephedrine (Problem 6.31) about the C1C2 bond for all three staggered and all three eclipsed conformations. (b) Examine each conformation for chirality. How do the...
-
The difference in the standard free energies of formation for 1-butene and 2-methylpropene is 13.4 kJ mol 1 (3.2 kcal mol 1 ). (See the previous problem for a definition of G f .) (a) Which compound...
-
Marshall uses a job order costing system to account for projects. It applies manufacturing overhead to jobs on the basis of direct labor hours and pays its direct labor workers $30 per hour. The...
-
Brembo, an Italian automotive braking-systems supplier, announced that it was going to split the stock 5-for-1. Brambilla Fumagalli owns 100 shares of the company, which he had purchased for 40 per...
-
The Motor Corporation sold vehicles for $500,000 to one specific dealer during the year. At the end of the financial year, the dealer still owed The Motor Corporation $350,000. The cost of the...
-
Dream Cakes bought some baking ingredients costing $12,500 on credit. The credit terms are 2.5/5 net 30 EOM. If Dream Cakes pays the invoice within 5 days after the purchase, how much should it pay?...
-
Barnes Limiteds most recent dividend was 2.20 per share, its expected annual rate of dividend growth is 5%, and the required return is 12%. Its management is considering a variety of proposals to...
-
There is a renewed interest in the extent to which government regulations can be incorporated in corporate governance and financial markets and institutions. There are various arguments both in favor...
-
Book value of an existing asset that we are contemplating replacing is nothing but a bookkeepers figure and, as such, is irrelevant in capital-expenditure analysis. Do you agree? Why or why not?
-
How do network effects help Facebook fend off smaller social-networking rivals? Could an online retailer doing half as much business compete on an equal footing with Amazon in terms of costs? Explain.
-
How might you synthesize each of the following compounds using an aldol reaction? Show the structure of the starting aldehydes(s) or ketone(s) you would use in eachcase. (a) (b) (c) (d) CeH5 CeH5 - ...
-
What product would you except to obtain from aldol cyclization of hexane-dial, OHCCH 2 CH 2 CH 2 CH 2 CHO?
-
Intermolecular aldol cyclization of 2, 5-heptanedione with aqueous NaOH yields a mixture of two enone products in the approximate ratio 9:1. Write their structures, and show how each is formed.
-
Celestila Moonn, a GLBAAF 301 student at UMB, made the following statements regarding preferred stock and capital gain: Statement 11: A long-term investor in a high marginal tax bracket will normally...
-
Exercise 3. Let y: [0, 1] R2 be the curve defined by y(t) = (2+t,t + 1). The line integral is equal to A2e-2. B 2e. et/y Ax+2 ds Ce+1. De-1.
-
5. Determine an expression for f(x) in which f(x) is a quartic function f(x) has a root of order 2 at x=-2 and two other roots at x=2 and x = 1 f(-1)= 14. [3K]

Study smarter with the SolutionInn App


