Predict which of the following compounds should undergo the more rapid reaction with K + (CH 3
Question:
Predict which of the following compounds should undergo the more rapid reaction with K+(CH3)3C—O¯, explain your reasoning, and give the product of the reaction.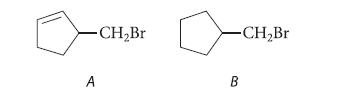
Transcribed Image Text:
-CH₂Br A -CH₂Br B
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 100% (5 reviews)
Alkyl halides with allylic or benzylic hydrogens undergo more rapid E2 ...View the full answer

Answered By

Utsab mitra
I have the expertise to deliver these subjects to college and higher-level students. The services would involve only solving assignments, homework help, and others.
I have experience in delivering these subjects for the last 6 years on a freelancing basis in different companies around the globe. I am CMA certified and CGMA UK. I have professional experience of 18 years in the industry involved in the manufacturing company and IT implementation experience of over 12 years.
I have delivered this help to students effortlessly, which is essential to give the students a good grade in their studies.
3.50+
2+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
Predict which of the following two compounds will undergo an E2 reaction more rapidly: CI
-
Predict which of the following compounds is more acidic. After making your prediction, use the pK a values from the following table to determine whether your prediction was correct. ACID pka...
-
Predict which of the following compounds is more acidic, and explain your choice. N- -N- -NH2 NH2
-
Write a program that takes an integer command-line argument n and creates an n-by-n boolean array a[][] such that a[i][j] is true if i and j are relatively prime (have no common factors), and false...
-
Interpreting the statement of cash flows Exhibit 5.37 presents statements of cash flows for Spokane Corporation, a forest products company, for three years from its past when it faced financial...
-
The executives of a company that produces electro-optical equipment for military use found it necessary to implement project management using a matrix. The project managers reported to corporate...
-
To charge the batteries in a Tesla Roadster, a transformer is used to step up the voltage of the household supply. If you step a \(220 \mathrm{~V}, 100\) A system up to \(400 \mathrm{~V}\), what is...
-
Nazir Khan, the CEO of Peshawar Inc., signed an employment contract with the company that allowed him to earn a bonus if he increased Peshawar's gross profit margin by more than 3%. The draft income...
-
Delph Company uses a job-order costing system and has two manufacturing departments-Molding and Fabrication. The company provided the following estimates at the beginning of the year: Machine-hours...
-
A hydrocarbon A, C 9 H 10 , is treated with N-bromosuccinimide to give a single monobromo compound B. When B is dissolved in aqueous acetone it reacts to give two nonisomeric compounds: C and D....
-
Terfenadine is an antihistaminic drug that contains two alcohol functional groups. Suppose terfenadine were to undergo acid-catalyzed alcohol dehydration (Sec. 10.2). Which alcohol would dehydrate...
-
The pipe of mass M is supported at A by a system of five cords. Determine the force in each cord for equilibrium. Given: M = 30 kg c = 3 g = 9.81 m/s2 d = 4 θ = 60 deg B E
-
What is the total tax due for 2019, including self- employment tax, for Stuart, assuming that he earned $20,000 in wages, earned $24,000 in self-employment income from his first business, had a loss...
-
Refer to a citator and locate the following case: New York Life Insurance Co. v. Edwards. a. What happened on appeal to the Court of Appeals? b. What happened on appeal to the Supreme Court?
-
Joe signed for a ten-year lease to rent office space from Stanley. In the first year, Joe paid Stanley $5,000 for the first years rent and $5,000 as rent for the last year oft he lease How much must...
-
Helen Troy, a student seeking a Master's degree in accounting, was awarded a 50 percent scholarship to graduate school (valued at $3,000) and a $6,000 per year teaching assistantship (not required of...
-
What Code Section immediately follows Code Sec. 280?
-
Using the results of problem, compute a critical value by using the Tukey-Kramer procedure for groups 1 and 2. Use = .05. Determine whether there is a significant difference between these two...
-
What key concerns must functional tactics address in marketing? Finance? POM? Personnel?
-
Carry out an analysis similar to Study Problem 11.6 of the stereochemical result expected when the (2R,3R)-stereoisomer of the starting material is used.
-
Outline a synthesis for each of the following compounds from the indicated starting materials and any other reagents: (a) (CH3)2CHCOTH from 2-methylpropene (b) Dibutylsulfone from l-butanethiol
-
Would the same phenomenon be observed with (-)-DET, the enantiomer of the DET used in Fig. I 1.2? Explain.
-
A person standing on the edge of a high cliff throws a rock straight up with an initial velocity of 15.4 m/s. The rock misses the edge of the cliff as it falls back to earth. Let y = 0, where y, is...
-
Problem 2. Impulse graph. This is a graph of the x-component of a force on an object. The functional form of the red curve is F x t, and the form of the blue curve is F xx et. The symbol x means...
-
Problem 1. Conservation debate. Two friends are having a friendly disagreement on the conservation of energy and momentum. "Conservation of energy and momentum are basic principles of physics. They...

Study smarter with the SolutionInn App


