When the following compound is treated with H 2 SO4, the product of the resulting reaction has
Question:
When the following compound is treated with H2SO4, the product of the resulting reaction has the formula C15H20 and does not decolorize Br2 in CCl4. Suggest a structure for this product and give a curved-arrow mechanism for its formation.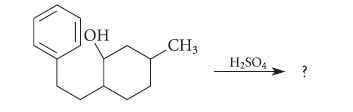
Transcribed Image Text:
он CH3 H2SO4 ?
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 75% (4 reviews)
The product has an unsaturation number of 6 which means that relative to the starting mate...View the full answer

Answered By

Joram mutua
I am that writer who gives his best for my student/client. Anything i do, i give my best. I have tutored for the last five years and non of my student has ever failed, they all come back thanking me for the best grades. I have a degree in economics, but i have written academic papers for various disciplines due to top-notch research Skills.In additional, I am a professional copywriter and proofreader.
5.00+
2+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
When the following compound is treated with Br 2 in the presence of a Lewis acid, one product predominates. Determine the structure of that product. Br FeBr3 ?
-
When the following compound is treated with sodium methoxide in methanol, two elimination products are possible. Explain why the deuterated product predominates by about a 7:1 ratio (refer to Problem...
-
When the following compound is treated with a mixture of nitric and sulfuric acid at 50°C, nitration occurs to afford a compound with two nitro groups. Draw the structure of this product:...
-
For students in the United States, compare Medicaid coverage in your state with coverage afforded to recipients in a neighboring state. Are they the same? If not, why do you think that they differ?
-
The statement of cash flows classifies changes in accounts payable as an operating activity but classifies changes in short-term bank borrowing as a financing activity. Explain this apparent paradox.
-
Pierot Theater Company's trial balance at the end of its current fiscal year is shown below. Required 1. Enter Pierot Theater Company's trial balance amounts in the Trial Balance columns of a work...
-
In years past, car frames were built as stiff as possible, but today's cars are built with "crumple zones" in the front and rear portions of the frame. What is the purpose of this design change?
-
A researcher took an SRS of 4 high schools from a region with 29 high schools for a study on the prevalence of smoking among female high school students in the region. The results were as follows: a....
-
Chocolaterie de Geneve, SA, is located in a French-speaking canton In Switzerland. The company makes chocolate truffles that are sold in popular embossed tins. The company has two processing...
-
Diphenylsulfone is a by-product that is formed in the sulfonation of benzene. Give a curved-arrow mechanism for its formation. oto diphenylsulfone
-
Two alcohols, A and B, have the same molecular formula C 9 H 10 O and react with sulfuric acid to give the same hydrocarbon C. Compound A is optically active, and compound B is not. Catalytic...
-
Evaluate the following integrals. [cos cos x V sin x dx
-
Frank and Emma Browne are both over 65 and file a joint return. Emma received $800 from Social Security benefits and Frank received $1,200 from railroad retirement benefits. In addition to the...
-
Norma, a single taxpayer, had 2019 taxable income before the qualified business income deduction of $650,000, of which $500,000 was qualified business income earned through her sole proprietorship....
-
Larry Johnson, 45 and single, has the following income and deductions in 2019. Using the tax rate schedules rather than the tax tables, compute Larrys tax owed or refund due for 2019. Assume...
-
Compute the child and dependent care credit in each of the following independent cases: a. Jack and Jill Jones are married and file a joint return. Jill worked full time earning $20,000, while Jack...
-
During the following years Adolpho T. (single) had (1) withheld, (2) paid (by check presumably estimated tax payments or paid with the extension) and (3) received a refund of the following amounts...
-
One of the most important aspects of a store's image is the perceived quality of its merchandise. Other factors include merchandise pricing, assortment of products, convenience of location, and...
-
Dan and Diana file a joint return. Dan earned $31,000 during the year before losing his job. Diana received Social Security benefits of $5,000. a. Determine the taxable portion of the Social Security...
-
A student consulted a well-known compilation of reference spectra for the proton NMR spectrum of propyne and was surprised to find that this spectrum consists of a single unsplit resonance at 51.8....
-
The addition of HCI to 3-hexyne occurs as an anti-addition. Give the structure, stereochemistry, and name of the product.
-
Give the diere and dienophile that would react in a Diels-Alderreaction to give each of the following products. CN
-
3. If tolerable misstatement as percentage of 630,000 population value is 5% and and Confidence factor is 3.68 Calculate sampling interval (1 Point) 8560 31500 63.7 73.6 4. Increasing ARIA leading to...
-
List 3 factors that officers rely on to evaluate H&C applications.?
-
On its income statement for the year, Crescent Ltd. reported $100,000 of selling and administrative expenses, which included $20,000 in depreciation expense. During the year prepaid expenses...

Study smarter with the SolutionInn App


