When the following compound is treated with a mixture of nitric and sulfuric acid at 50°C, nitration
Question:
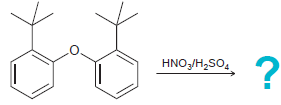
Transcribed Image Text:
HNO3/H,SO.
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 80% (5 reviews)
HNO...View the full answer

Answered By

Mugdha Sisodiya
My self Mugdha Sisodiya from Chhattisgarh India. I have completed my Bachelors degree in 2015 and My Master in Commerce degree in 2016. I am having expertise in Management, Cost and Finance Accounts. Further I have completed my Chartered Accountant and working as a Professional.
Since 2012 I am providing home tutions.
3.30+
2+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
What product is formed when the following compound is treated with Ag2O? HO
-
When the following compound is treated with sodium methoxide in methanol, two elimination products are possible. Explain why the deuterated product predominates by about a 7:1 ratio (refer to Problem...
-
Only a substitution product is obtained when the following compound is treated with sodium methoxide: Explain why an elimination product is not obtained. CH3 Br CH3
-
The following is a summary of the petty cash transactions of Jockfield Ltd for May 2012. You are required to: (a) Rule up a suitable petty cash book with analysis columns for expenditure on cleaning,...
-
What is your evaluation of Starbucks social responsibility strategy? Is it sincere or just something the company does and talks about to create a good public image? What grade would you give Howard...
-
For the Catheter Study, the patients were assessed bimonthly and the measurements about UTIs, catheter blockages, and replacements cover the previous two months. Thus, the patients were under...
-
Provide three examples for open-ended and closed questions.
-
The trial balance and adjustments columns of the worksheet of Budget Business Systems at March 31, 2012, follow: Requirements 1. Compute the adjusted balance for each account that must be closed. 2....
-
How does one reconcile ethical dilemmas in professional settings, particularly when they intersect with personal values and societal norms ?
-
Silver Lining, Inc., provides investment advisory services. The company adjusts its accounts monthly, but performs closing entries annually on December 31. The firms unadjusted trial balance dated...
-
Predict the major product obtained when each of the following compounds is treated with bromine in the presence of iron tribromide. (a) Bromobenzene (b) Nitrobenzene (c) ortho-Xylene (d)...
-
When benzene is treated with 2-methylpropene and sulfuric acid, the product obtained is tert butylbenzene. Propose a mechanism for this transformation.
-
Which WAN technology is a cost-effective solution for connecting multiple locations? A. MPLS B. ISDN C. MAN D. L2TP
-
Suppose that John Smith gets promoted to a job that causes two changes to occur simultaneously: John earns a higher wage, and a safer environment causes his health to depreciate less rapidly. How...
-
Identify fi ve distinctive features of the health economy. Examine each one separately, and describe other commodities or sectors that share those features. Do any other commodities or sectors have...
-
A Mudgee Ltd issued the following invoices to customers in respect of credit sales made during the last week of May 2013. The amounts stated are all net of Value Added Tax. All sales made by Mudgee...
-
A D. Faculti started in business buying and selling law textbooks, on 1 January 2012. At the end of each of the next three years, his figures for accounts receivable, before writing-off any bad...
-
From the following figures, compile accounts receivable ledger and accounts payable ledger control accounts for the month, and ascertain what the net balances of the respective ledgers should be on...
-
Draw an ASM chart for the following: Assume three states (a, b, c) in the system with one input x and two registers R 1 and R 2 . The circuit is initially in state a. If x = 0, the control goes from...
-
After Theorem 1.5 we note that multiplying a row by 0 is not allowed because that could change a solution set. Give an example of a system with solution set S0 where after multiplying a row by 0 the...
-
Epoxy adhesives are prepared in two steps. SN2 reaction of the disodium salt of bisphenol A with epichiorohydrin forms a ?prepolymer,? which is then ?cured? by treatment with a triamine such as H 2...
-
In the iodoform reaction, a triiodomethyl ketone reacts with aqueous NaOH to yield a carboxylate ion and iodoform (triiodomethane). Propose a mechanism for thisreaction. OH H20 Cl3 HCI3 R.
-
Draw structures for the enol tautomers of the following compounds: (a) Cyclopentanone (b) Methyl thioacetate (c) Ethyl acetate (d) Propanal (e) Acetic acid (f) Phenyl acetone
-
Create a segmentation, selection and focus model that allows the PUMA company in Mexico to determine its customer, where to find it and how to generate value given its model. 7) Selection of the most...
-
Determine if the following values are zeroes polynomial. 4 3 f(x) = x + x-18x - 16x + 32 x+1, +2, +4 -
-
Cali Windows is a small company that installs windows. Its cost structure is as follows: (Click the icon to view the cost structure.) Calculate (a) the breakeven point in units and revenues and (b)...

Study smarter with the SolutionInn App


