Which of the following are Fischer projections of a meso compound? - - - CH-0 - -OH
Question:
Which of the following are Fischer projections of a meso compound? 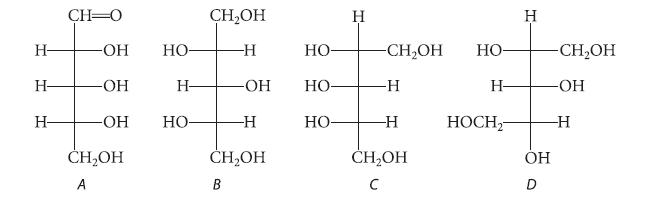
Transcribed Image Text:
Н- Н- Н- CH-0 -ОН -OH -ОН CH₂OH A НО Н- но- CH₂OH -Н -OH -Н CH₂OH B но- но- НО- Н -CH₂OH -Н -Н CH₂OH с НО- Н- HOCH₂ Н ОН -CH₂OH -ОН -Н
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 33% (3 reviews)
It is relatively simple to determine if a compound is meso using Fisher projections First perform a ...View the full answer

Answered By

Ujjwal Ghosh
I have been teaching for last 10 years. Many students whom I've taught are now studying the courses of their choice. Most of my students cracked the competitive examinations in the first attempt.
I've taught many students from graduation to higher level like master degree courses.
They were very happy with my teaching style.
I can solve higher order thinking questions in very less time.
https://www.linkedin.com/in/ujjwal-ghosh-1a506a1b4/
0.00
0 Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
Following are Fischer projections for a group of five-carbon sugars, all of which are aldopentoses. Identify the pairs that are enantiomers and the pairs that are epimers. (The sugars shown here are...
-
Which of the following compounds are chiral? Draw each compound in its most symmetric conformation, star (*) any asymmetric carbon atoms, and draw any mirror planes. Label any meso compounds. You may...
-
Which compound is not a meso compound? (a) (b) (c) (d) (e) Cl Cl CH3 Cl Cl Cl
-
An array is bitonic if it consists of an increasing sequence of keys followed immediately by a decreasing sequence of keys. Given a bitonic array, design a logarithmic algorithm to find the index of...
-
The K 0 particle has a rest mass of 497.7 MeV/c 2 . It decays into a and + , each with rest mass 139.6 MeV/c 2 . Following the decay of a K 0 , one of the pions is at rest in the laboratory....
-
The marketing department of Metroline Manufacturing estimates that its sales in 2013 will be $1.5 million. Interest expense is expected to remain unchanged at $35,000, and the firm plans to pay...
-
The air in a hot-air balloon can be heated only so much; the temperature of the gas inside the balloon can only be so high before damaging the nylon envelope. This means that hot-air balloons can...
-
On January 2, 2014, Placer Company acquired a 75 percent interest in Summer Company for $8,000,000 in cash. The condensed balance sheets immediately prior to the Acquisition are below: Additional...
-
WhStudents will be able to convert numbers by using the "making 10" strategy when adding and subtracting problems within 20.at is a specific example of a formal-formative assessment using NY2OA2a and...
-
Which pair of the following aldoses are epimers and which pair are enantiomers? H HO H CH=0 -OH A - -OH -CHOH 0=CH- - OH H B - -H -OH -CHOH H - OH -CH=0 -OH - - CHOH
-
Name each of the following aldoses. In part (a), work back to the Fischer projection and consult Fig. 24.3. In part (b), decide which carbons have configurations different from those of glucose, and...
-
Use the data in Exercise 9.39 to calculate the annual percentage change in income and consumption, for example, in 1961 a. Draw a scatter diagram using these annual percentage changes. b. Estimate...
-
Explain how the desired levels of capital and investment are affected by changes in the expected marginal product of capital, the user cost of capital, and taxes.
-
What objections to the real business cycle model have been raised?
-
One of the possible solutions to asset-price bubbles is the enforcement of macroprudential regulation. Financial intermediaries have an incentive to constantly look for profitable opportunities,...
-
How do the traditional Keynesian, new Keynesian, and real business cycle models differ in their analysis of the effects of expansionary policy?
-
What is the significance of the Lucas critique of econometric policy evaluation?
-
Suppose the mean clear height of all U.S. Class A warehouses is unknown but the standard deviation is known to be 4 feet.What is the value of the mean clear height if 29% of U.S. Class A warehouses...
-
Explain the circumstances that could result in a long-term bank loan being shown in a statement of financial position as a current liability.
-
Circle any lone pairs (pairs of nonbonding electrons) in the structures you drew for Problem 1-3. In problem (a) N2 (b) HCN (c) HONO (d) CO2 (e) CH3CHNH (f) HCO2H (g) C2H3CI (h) HNNH
-
In the following sets of resonance forms, label the major and minor contributors and state which structures would be of equal energy. Add any missing resonance forms. (a) (b) (c) (d) (e) CH,_...
-
For each pair of ions, determine which ion is more stable. Use resonance forms to explain your answers. (a) (b) (c) (d) (e) CH CHCH or CH CH OCH CH CH CH-CH or CH CH CH2 CH CH,_ CH, or CH,-C N: CH2...
-
How do spatial and temporal variations in resource availability, such as seasonality, habitat heterogeneity, and nutrient gradients, influence food chain dynamics, including predator-prey...
-
1 . A semiannual - pay bond has 7 years to maturity, a yield to maturity of 7 . 4 1 percent and is priced at $ 1 , 0 1 2 . 6 7 . What is its annual coupon rate? Answer in percent to two decimals. 2 ....
-
Many people mention responding to emails and voice messages within 24 hours. Why do you feel this is important, and how do you believe it affects the recipient when messages are not returned in a...

Study smarter with the SolutionInn App


