Explain why 1-methylaziridine undergoes amine inversion much more slowly than 1-methylpyrrolidine. (What are the hybridization and bond
Question:
Explain why 1-methylaziridine undergoes amine inversion much more slowly than 1-methylpyrrolidine. (What are the hybridization and bond angles at nitrogen in the transition state for inversion?)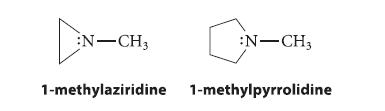
Transcribed Image Text:
N-CH3 :N-CH3 1-methylaziridine 1-methylpyrrolidine
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 50% (2 reviews)
In the transition state for inversion of both amines the nitrogen is sphybridized The CNC ang...View the full answer

Answered By

Ali Khawaja
my expertise are as follows: financial accounting : - journal entries - financial statements including balance sheet, profit & loss account, cash flow statement & statement of changes in equity -consolidated statement of financial position. -ratio analysis -depreciation methods -accounting concepts -understanding and application of all international financial reporting standards (ifrs) -international accounting standards (ias) -etc business analysis : -business strategy -strategic choices -business processes -e-business -e-marketing -project management -finance -hrm financial management : -project appraisal -capital budgeting -net present value (npv) -internal rate of return (irr) -net present value(npv) -payback period -strategic position -strategic choices -information technology -project management -finance -human resource management auditing: -internal audit -external audit -substantive procedures -analytic procedures -designing and assessment of internal controls -developing the flow charts & data flow diagrams -audit reports -engagement letter -materiality economics: -micro -macro -game theory -econometric -mathematical application in economics -empirical macroeconomics -international trade -international political economy -monetary theory and policy -public economics ,business law, and all regarding commerce
4.00+
1+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
Pyramidal inversion in the cyclic amine aziridine is significantly more difficult than inversion in an acyclic amine; for example, requiring 80 kJ/mol versus 23 kJ/mol in dimethylamine according to...
-
Using the following information on Rockboro Case please answer the following Question: What are the problems here, and what do you recommend and what are the implications of different payout levels...
-
1. Draw the important resonance forms for each compound. 2. Label the hybridization and bond angles around each atom other than hydrogen. 3. Use a three-dimensional drawing to show where the...
-
The following question is designed to highlight key concepts from the Loyalty Programs topic article titled, "StarBUCKS, Loyalty, and Breakage" (Nevraumont 2019). Q. Author's position. "If you hire...
-
Talk to five people, varying in age from young adult to senior citizen, about their automobiles. Ask them what value means to them with regard to an automobile and how the manufacturer and dealer...
-
Access the website for CVS Corporation and locate the companys 2005 annual report. Conduct a comprehensive ratio analysis that compares the companys performance in 2005 and 2004. If you have computed...
-
Briefly explain how the following programs would affect the elasticity of demand for labor in the steel industry: a. An increased tariff on steel imports. b. A law making it illegal to lay off...
-
Gershwin Corporation obtained a franchise from Sonic Hedgehog Inc. for a cash payment of $120,000 on April 1, 2010. The franchise grants Gershwin the right to sell certain products and services for a...
-
In some labor markets, the supply of labor is as follows (where W represents wages and L represents the number of employees): W = 113 + 0.21L However, the demand for labor is: W = 958 -0.28L a) At...
-
Alkaline potassium permanganate (KMnO 4 ) can be used to bring about the addition of two OH groups to an alkene double bond. This reaction has been shown in several cases to be a stereospecific...
-
Which of the following compounds can be resolved into enantiomers at room temperature? Explain. (a) Z: (c) HC- CH3 CH3 CH3 (b) CHCH3 T CH3 CH3 (d) CHCH3 - CH3 H
-
In Exercises find the nth Maclaurin polynomial for the function. (x) = e -x , n = 5
-
Find the following amounts in the statements: (Enter amounts in thousands. Round Gross profit as a percentage in 2020 to 1 decim place, e.g. 15.2%.) Revenue in 2020 ii. Cost of sales in 2020 iii....
-
Unix Operating Systems and User Interface ----------------------------------------------------------------------------------------------------------------- Q. Mobile Operating System Research and...
-
There are many different operating systems that you might encounter while working in the healthcare industry. Different types of computers, tablets, and phones use different operating systems for...
-
1. You are a financial planner. You are invited to speak at a conference of varying investors. What they share in common is they don't believe in diversifying their investments. What they don't have...
-
Skhumbuzo is a private business man who began his trade by operating a spaza shop in the vicinity of the University of Eswatini in the area of Kwaluseni. His business has a special focus on three hot...
-
A 20-mm-diameter steel bar is to be used as a torsion spring. If the torsional stress in the bar is not to exceed 110 MPa when one end is twisted through an angle of 15, what must be the length of...
-
Suppose the index goes to 18 percent in year 5. What is the effective cost of the unrestricted ARM?
-
Consider the free energy versus reaction progress diagram for the SN2 reaction shown in Figure 8.1. Does the transition state for this reaction have the C Cl bond less than half broke, approximately...
-
Explain which compound has a faster rate of SN1 reaction. a) c) HC CI CI or or J d) CI CHCI or or D CHCl OCH 3
-
Arrange these compounds in order of decreasing SN1 reaction rate. Ph CI CI CI Ph CI Ph
-
(a) What is the concentration of hydrogen at the B face in kilograms of H per cubic meter? C"H(B) == i kg/m (b) What is the concentration of hydrogen at the A face in kilograms of H per cubic meter?...
-
Selected accounts of Wildhorse Company are shown as follows. Supplies Expense 7/31 800 Supplies 7/1 Bal. 1,300 7/31 800 7/10 550 Accounts Receivable 7/31 500 Salaries and Wages Expense 7/15 1,500...
-
Propose the structure of the product formed in each of the following reactions. (syntesis) . HCHO Product A HCI H,C 0 NaOH) Product H + H.C PH2 COOH DMF / POCI 3 Product "N" H CH CH HNO AcO Product...

Study smarter with the SolutionInn App


