Which of the following compounds should have the larger energy barrier to internal rotation about the indicated
Question:
Which of the following compounds should have the larger energy barrier to internal rotation about the indicated bond?
Explain your reasoning carefully.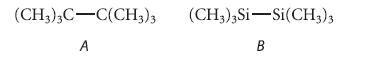
Transcribed Image Text:
(CH3)3C-C(CH3)3 (CH3)3 Si-Si(CH3)3 A B
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 100% (1 review)
A major contributor to the barrier to internal rotation is the van de...View the full answer

Answered By

Jeff Omollo
As an educator I have had the opportunity to work with students of all ages and backgrounds. Throughout my career, I have developed a teaching style that encourages student engagement and promotes active learning. My education and tutoring skills has enabled me to empower students to become lifelong learners.
5.00+
5+ Reviews
42+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
From what you learned in Sec. 1.3B about the relative lengths of CC and CO bonds, predict which of the following compounds should have the larger energy difference between gauche and anti...
-
KYC's stock price can go up by 15 percent every year, or down by 10 percent. Both outcomes are equally likely. The risk free rate is 5 percent, and the current stock price of KYC is 100. (a) Price a...
-
Sketch a diagram of potential energy versus angle of rotation about the carboncarbon bond of chloroethane, H 3 CCH 2 Cl. The magnitude of the energy barrier to internal rotation is 15.5 kJ mol 1 (3.7...
-
Consider the many moments of joy in the movie. Why include them? What risky acts of courage do Katherine, Mary and Dorothy take? How are they rewarded? The film shows the strong relationships that...
-
The court seems to regard Joness allegations as trivial. In fact, hasnt she alleged disgusting behavior by her employer? How can the court regard her claims so lightly?
-
Heartwood Furniture Corporation has a line of sofas marketed under the name NightTime Sleepers. Heartwood management is considering several compensation packages for Amy Johnson, NightTime's general...
-
Tandrin Aviation Holdings Ltd. agreed to sell a jet aircraft to Aero Toy Store, LLC, for \($31.75\) million. ATS paid a \($3\) million deposit to a third party with the balance due upon delivery....
-
The following data relate to the Machinery account of Eshkol, Inc. at December 31, 2012. *In the year an asset is purchased, Eshkol, Inc. does not record any depreciation expense on the asset.In the...
-
a) Simplify the expression. Express your answer with positive exponents. (4x6) (4) (2x)* (123) 2 b) In 2004, a sum of $1900 is invested at a rate of 3.75% per year (compound interest) for 12 years....
-
(a) Draw Newman projections of the most stable conformations about each of the carboncarbon bonds in the principal chain of 2,2-dimethylpentane. Use models! (b) Combine these to predict the most...
-
The anti conformation of 1,2-dichloroethane, ClCH 2 CH 2 Cl, is 4.81 kJ mol1 (1.15 kcal mol 1 ) more stable than the gauche conformation. The two energy barriers (measured relative to the energy of...
-
In designing a relational database, why might we choose a non-BCNF design?
-
check the following blog, and help with the following questions. https://www.hubspot.com/products/marketing/marketing-automation-information Questions Have you had experience with marketing...
-
Why should you create an account with Replit? To use all of the features of Replit. To run any Java application. To be able to compile Java applications. To make use of files in Replit.
-
In the early 1980s, the Reagan administration had very tight monetary policy (decrease in money supply), a big tax cut, and a big increase in government spending. The net effect was an increase in...
-
You have 30% of your investment in a stock with a beta of 1.5 and the rest in a stock with a beta of 1.1. The expected market return is 6.5% and the risk-free rate is 1%. What's the expected return...
-
In order to build a synergistic team, you need to build relationship with other team members. You are in the middle of the COVID-19 pandemic. You are the building representative on the homeowner's...
-
A. If consumption increases by $12 billion when real disposable income increases by $15 billion, what is the value of the MPC? B. What is the relationship between the MPC and the MPS? C. If the MPC...
-
Suppose that you are part of a virtual team and must persuade other team members on an important matter (such as switching suppliers or altering the project deadline). Assuming that you cannot visit...
-
When glucose (Problem) is treated with NaBH4, reaction occurs to yield sorbitol, a polyalcohol commonly used as a food additive. Show how this reductionoccurs. CH- NABHA -2 Sorbitol Glucose
-
Give IUPAC names for the followingcompounds: (a) Br (b) (c) CH CH3CH2CHCH2CH2CH3 CHH-CH2Co CHCH2C (f) O2- (d) CH CN (e) CHCCH2CHH "Co2 CH2CH2COH
-
Draw structures corresponding to the following IUPAC names: (a) 2, 3-Dimethylhexanoic acid (b) 4-Methylpentanoic acid (c) Trans-1, 2-Cyclobutanedicarboxylic acid (d) o-Hydroxybenzoic acid (e) (9Z,...
-
Test the following polar equations for symmetry with respect to the line 0 = /2, the polar axis and the pole. A. r = -3sin(20) B. r2 1 2cos(0) Remember: cos(-0) = cos(0) sin(-0) = -sin(0) Write the...
-
1. If a certain spring is stretched 2 m, it will produce a force of 10 N. Set up an integral to find the amount of work done in stretching the spring an additional 3 m.
-
The graph below is the function f(x) 00 4 Find lim f(x) x-3 Find lim f(x) x-3+ Find lim f(x) x 3 Find f(3)

Study smarter with the SolutionInn App


