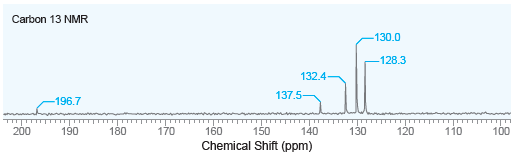
A compound with molecular formula C 13 H 10 O produces a strong signal at 1660 cm
Question:
Transcribed Image Text:
Carbon 13 NMR -130.0 -128.3 132.4- 137.5- -196.7 200 180 170 160 150 Chemical Shift (ppm) 140 130 120 110 100 190
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 54% (11 reviews)

Answered By

Mario Alvarez
I teach Statistics and Probability for students of my university ( Univerisity Centroamerican Jose Simeon Canas) in my free time and when students ask for me, I prepare and teach students that are in courses of Statistics and Probability. Also I teach students of the University Francisco Gavidia and Universidad of El Salvador that need help in some topics about Statistics, Probability, Math, Calculus. I love teaching Statistics and Probability! Why me?
** I have experience in Statistics and Probability topics for middle school, high school and university.
** I always want to share my knowledge with my students and have a great relationship with them.
** I have experience working with students online.
** I am very patient with my students and highly committed with them
5.00+
1+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
A compound with molecular formula C 9 H 10 O exhibits the following spectra ( 1 H NMR, 13 C NMR, and IR). Identify the structure of this compound.. Proton NMR 10 Chemical Shift (ppm) Carbon NMR 128.5...
-
The IR spectrum of a compound with molecular formula C5H8O was obtained in CCl4 and is shown in Figure 13.42. Identify the compound. Wavelenga qum) 15 16 14 3600 340) 3800 3300 3000 280K 2600 2400...
-
A compound with molecular formula C 8 H 10 O produces six signals in its 13 C NMR spectrum and exhibits the following 1 H NMR spectrum. Deduce the structure of the compound. Proton NMR Chemical Shift...
-
The table shows the fees for refund anticipation loans (RALs) offered by an online tax preparation firm. Find the annual rate of interest for each of the following loans. Assume a 360-day year. (A) A...
-
Pete is considering placing a bet on the NCAA playoff game between Indiana and Purdue. Without any further information, he believes that each team has an equal chance to win. If he wins the bet, he...
-
Consider scattering onto a delta function potential, \(V=-V_{0} \delta^{3}(\vec{r})\), in the Born approximation. Calculate the partial wave amplitudes \(a_{l}(k)\).
-
Students applying to graduate schools in many disciplines are required to take the Graduate Record Examination (GRE); essentially, it is the graduate school equivalent of the SAT. Let's say an...
-
Natherson, CPA, is engaged to audit the financial statements of Lewis Lumber for the year ended December 31. Natherson obtained and documented an understanding of internal control relating to the...
-
An online book retailer, has two operating divisionslong dashcorporate sales and consumer saleslong dashand two support divisionslong dashhuman resources and information systems. Each sales division...
-
Which of the graphs in Fig. Q25.12 best illustrates the current I in a real resistor as a function of the potential difference V across it? Explain. Figure Q25.12 (a) (b) (c) (d)
-
A compound with molecular formula C 9 H 10 O exhibits a strong signal at 1687 cm 1 in its IR spectrum. The 1 H and 13 C NMR spectra for this compound are shown below. Identify the structure of this...
-
A ketone with molecular formula C 9 H 18 O exhibits only one signal in its 1 H NMR spectrum. Provide a systematic (IUPAC) name for this compound.
-
If the fourth oil change and lube job at Trendo-Lube took 18 minutes and the second took 20 minutes, estimate how long a) The first job took. b) The third job took. c) The eighth job will take. d)...
-
Class Activity 10: Persuasive Sales Letter You are a marketing manager of a large size software house. Through some reliable sources, you have got information that Saputo Dairy Foods Canada is not...
-
Perspectives on the role of media in society Watch the entire discussion on YouTube between Lars Ulrich, Chuck D, and Charlie Rose:Lars Ulrich, Chuck D And Charlie Rose On Napster In 2000 Be sure to...
-
scenario ; we need extension! here is the situation. you are a student in the university. you are taking five classes and are quite busy between school and work commitments. One of your courses is...
-
Develop an optimization model involving product mix and transportation. Product is manufactured at A and B. A has capacity to produce 10 gallons. A requires 0.5 hour per gallon to manufacture...
-
How have the changes in currency markets impacted the returns in the last year and how may returns be impacted in the future? Why is it either a good idea or bad idea for U.S. investors to own stocks...
-
At the Virgin Music store in Times Square, 60 people entering the store were selected at random and were asked to choose their favorite type of music. Of the 60, 12 chose rock, 16 chose country, 8...
-
Use the graphs of f and g to graph h(x) = (f + g) (x). To print an enlarged copy of the graph, go to MathGraphs.com. 1. 2. y 24 8. 2. -2 -2 4 6
-
Rank the following alcohols in order of increasing ease of acid-catalyzed dehydration. OH OH OH
-
Acid-catalyzed dehydration of neopentyl alcohol, (CH3)3CCH2OH, yields 2-methyl-2- butene as the major product. Outline a mechanism showing all steps in its formation.
-
Acid-catalyzed dehydration of either 2-methyl-1-butanol or 3-methyl-1-butanol gives 2-methyl-2-butene as the major product. Write plausible mechanisms that explain these results.
-
Reflect on a group interview you've had: * Was there anything you struggled with in preparing for the peer interviews? * What would you have changed about your interview? What would you have kept the...
-
Please conduct comprehensive research on the status of Metaverse, Choose a company, an industry, a product, or a service, and Share the strategies being used in Metaverse to develop a competitive...
-
Renovation and Restoration of community Park project For Risk management plan External risk : Budget Fluctuations in local economy Environment disasters Internal Risk: Safety Not enough money, staff...

Study smarter with the SolutionInn App


