All of the following compounds absorb IR radiation in the range between 1600 and 1850 cm -1
Question:
a.
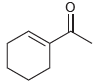
b.
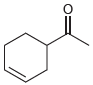
c.
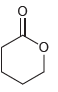
d.
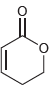
e.
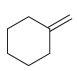
Transcribed Image Text:
O.
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 100% (7 reviews)
a b c...View the full answer

Answered By

Gaurav Soni
Myself a student of Physics currently pursuing my PG. I helped many students due to my understanding specifically among students and also in my subject i. e. Physics. Helping them in various boards and in various entrance examination gives me a satisfaction of gaining knowledge. I have taught physics to students for many engineering and medical entrance examination and also for various boards in india. Till now have a great results from my students and that's why having a good impact on them. I have a good knowledge in conceptual and theoretical physics. Essential University Physics is one of my favorite field of teaching. I have a experience of almost 4 years in teaching.
0.00
0 Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
All of the following compounds absorb infrared radiation between 1600 and In each case, 1. Show which bonds absorb in this region. 2. Predict the approximate absorption frequencies. 3. Predict which...
-
All of the following compounds can react as acids. Without using a table of acidities, rank them in order of increasing acidity. Explain your ranking. (a) CH3CH2SO3H (b) CH3CH2OH (c) CH3CH2COOH (d)...
-
What features will be similar in the IR spectra of the following compounds, and how will their IR spectra differ? H2C CHCHCH and CH CH CHCH,CHCH sDh
-
Find any horizontal or vertical asymptotes. f(x) = = X 1- X
-
The article by Gage-Bouchard discusses how social inequality manifests in health care settings, drawing from research on cultural health capital, fundamental cause theory and doctor-patient...
-
Assume the Lagrangian density (17.14) with \(i=1,2,3\) for an isovector Lorentz scalar field to be invariant under a global internal \(\mathrm{SO}(3)\) symmetry. Show that for \(\mu^{2} Data from Eq....
-
With reference to Exercise 4.5, find an expression for the distribution function \(F(x)\) of the random variable. Data From Exercise 4.5 k 4.5 Given that f(x): = is a probability distribution for 2x...
-
Multiple Choice Questions 1. The moving average inventory cost flow method is applicable to which of the following inventory systems? Periodic Perpetual a. Yes..... Yes b. Yes..... No c. No..... No...
-
Pharoah Company uses a job order cost system in each of its three manufacturing departments. Manufacturing overhead is applied to jobs on the basis of direct labor cost in Department D, direct labor...
-
Before marketing new products nationally, companies often test them on samples of potential customers. Such tests have a known reliability. For a particular product type, a test will indicate success...
-
Does each of the following nucleophiles favor S N 2 or S N 1? a) b) c) (d) NaOH (e) NaCN SH
-
Rank each of the bonds identified in order of increasing wave number. R-CEN - N-H - -
-
Joie Corporation has been using a voucher system for several years and records invoices at gross. Prepare entries in the voucher register and check register for the following transactions: 201X Sept....
-
Explain briefly two reforms proposed to assist mid-tier audit firms compete with the Big Four firms in the large company audit market.
-
Given the powerful nature of project management software packages today, why should a project manager have to know how to construct a PERT chart or WBS?
-
How might lean apply to a service like an airline? A retailer? A university?
-
It is generally not appropriate to apply control charts to the same data that were used to derive the mean and limits. Why? What are the two possible outcomes if this is done, how likely is each, and...
-
Briefly explain four ways in which corporate responsibility issues may impact the statutory audit of companies financial statements.
-
If the strong nuclear force had a longer range than it does, what effect (if any) would that have on efforts to harness controlled fusion as an energy source?
-
Anna, a high school counselor, devised a program that integrates classroom learning with vocational training to help adolescents at risk for school dropouts stay in school and transition to work...
-
In each case, which group receives the higher priority? (a) (b) HC-CH (CH3)C
-
Which of the following two reactions should have the greatest ÎH° change? Why? (1) (2) C(CH3) (CH3)Q C(CH3) H C CH3 H3C CH3
-
Supply the curved-arrow notation for the acid-catalvzed isomerizatron.
-
Find the first derivative of the following equation: a. b. y=e3x y=e(x+1) d. y=125x+16-2x y=ln x y=ln (3x+x1) y=In (x+1)
-
The Cutting Department of Lasso Company has the following production and cost data for August. Production Costs 1. Started and completed 9,600 units. Beginning work in process $0 2. Started 2,300...
-
There is sufficient evidence to support the conclusion that a robust relationship exists between drugs and crime. Explain and discuss the studies that support this conclusion. Further explain and...

Study smarter with the SolutionInn App


