Consider the following sequence of reactions, and identify the structures of compounds A, B, and C. Mg
Question:
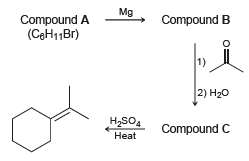
Transcribed Image Text:
Mg Compound A (CeH11Br) Compound B 1) 2) H20 H,SO4 Compound C Heat
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 83% (12 reviews)
Br Compound ...View the full answer

Answered By

Fahmin Arakkal
Tutoring and Contributing expert question and answers to teachers and students.
Primarily oversees the Heat and Mass Transfer contents presented on websites and blogs.
Responsible for Creating, Editing, Updating all contents related Chemical Engineering in
latex language
4.40+
8+ Reviews
22+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
Consider the following sequence of reactions, and identify the reagents ah. H. - ob-o - Br
-
Consider the following sequence of reactions: a) Explain how you could use IR spectroscopy to differentiate between compounds F and G. b) Explain how you could use IR spectroscopy to differentiate...
-
The herbicide trifluralin is prepared by the following sequence of reactions. Identify compound A and deduce the structure of trifluralin.
-
Consider a model of random interest rates R; between year i and i+1. The R, are i.i.d. random variables such that 1+ R; is LogNormal (0.03,0.0010) distributed. Suppose you deposit an amount of 4000...
-
Ocean Research of San Diego, California, just received a check in the amount of $800,000 from a customer in Bangor, Maine. If the firm processes the check in the normal manner, the funds will become...
-
Use the Goldman Medical Supplies data from P11-3B. The cash amounts for Interest Revenue, Salary Expense, Interest Expense, and Income Tax Expense are the same as the accrual amounts for these items....
-
A 70-kg soldier on a secret mission has to parachute from an airplane over the desert. It takes five seconds for him to release his parachute after he jumps. Taking the mass of the parachute as \(5...
-
The shareholders' equity section of Superior Corporations balance sheet as of December 31, 2015, is as follows: The following events occurred during 2016: Jan. 5 10,000 shares of authorized and...
-
Now we have a rod-shaped space station of length 1336 m and mass 6.24 x 10^6 kg, which can change its length (kind of like an old-fashioned telescope), without changing its overall mass. Suppose that...
-
A young woman named Seema (22) succumbed to injuries at a private hospital inGuwahati on 1st June, 2013 due to multiple organ failure as she had developedsevere health issues due to swallowing acid....
-
A gas is slightly above its Boyle temperature. Do you expect z to increase or decrease as P increases?
-
Using 2-propanol as your only source of carbon, show how you would prepare 2-methyl-2-pentanol.
-
List, in increasing order, the smallest 20 power products in R[x, y, z] for the order deglex with power products x m y n z s where z < y < x.
-
What are the two subcategories of proved developed reserves?
-
Suppose that the price of cut wood decreases, lowering the cost of furniture. What effect would this have on the U.S. furniture industry, in general, and on U.S. companies, in particular? Illustrate...
-
Can PRMS definitions be used by companies traded on the US stock exchanges?
-
German automobile manufacturer, Mercedes-Benz has repeatedly stated that it offers its customers a premium brand experience and doesnt just provide them with cars. Briefly explain what you can infer...
-
During analysis, the _______ are examined to find the _______ that will provide functionality for the system. (a) classes and use cases (b) use cases and classes (c) state diagrams and classes (d)...
-
Accidents are still occurring at the intersection of Academic Avenue and Bravado Boulevard. The football team is rushing into the intersection the moment light B turns green. They are colliding with...
-
Problem 3.5 (4 points). We will prove, in steps, that rank (L) = rank(LT) for any LE Rnxm (a) Prove that rank (L) = rank (LTL). (Hint: use Problem 3.4.) (b) Use part (a) to deduce that that rank(L) =...
-
Which of the following amino acids are more likely to be found on the outside of a globular protein, and which on the inside? Explain. (a) Valine (b) Aspartic acid (c) Phenylalanine (d) Lysine
-
The chloromethylated polystyrene resin used for Merrifield solid-phase pep- tide synthesis is prepared by treatment of polystyrene with chloromethyl methyl ether and a Lewis acid catalyst. Propose a...
-
An Fmoc protecting group can be removed from an amino acid by treatment with the amine base piperidine. Propose amechanism. pKg = 23 !! CH2 -NHCHCO- HoiHdo- NaOH H3NCHCO- Co2 2 Fmoc-protected amino...
-
Jamie Lee is attracted to the low monthly payment advertised for a vehicle lease. She may well be able to afford a more expensive car than she originally thought. Jamie Lee really needs to think this...
-
Explain virtual hosting and how to use either name-based or IP-based virtual hosting.?
-
Discuss the role of literary allusion in satire as a means of subverting conventions and critiquing societal norms.

Study smarter with the SolutionInn App


