The chloromethylated polystyrene resin used for Merrifield solid-phase pep- tide synthesis is prepared by treatment of polystyrene
Question:
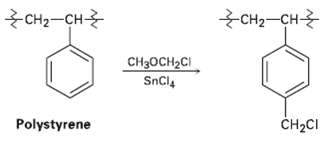
The chloromethylated polystyrene resin used for Merrifield solid-phase pep- tide synthesis is prepared by treatment of polystyrene with chloromethyl methyl ether and a Lewis acid catalyst. Propose a mechanism for thereaction.
Transcribed Image Text:
сHа-ҫн + сHа-сн Cн-оCH-CI SnCl4 Polystyrene CH2CI
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 92% (13 reviews)
Formation of cation HCCHC SnCl4 HCOCH SnC15 Ele...View the full answer

Answered By

RADHIKA MEENAKAR
I am a qualified indian Company Secretary along with Masters in finance with over 6 plus years of professional experience. Apart from this i am a certified accounts and finance tutor on many online platforms.
My Linkedin profile link is here https://www.linkedin.com/in/radhika-meenakar-88b9808a/
5.00+
12+ Reviews
22+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Organic Chemistry questions
-
The polymeric resin used for Merrifield solid-phase peptide synthesis (Section 26.8) is prepared by treating polystyrene with N-(hydroxymethyl) phthalimide and trifluoromethanesulfonic acid, followed...
-
The compound known as Hagemans ester is prepared by treatment of a mixture of formaldehyde and ethyl acetoacetate with base, followed by acid- catalyzed decarboxylation. (a) The first step is an...
-
Substituted pyrroles are often prepared by treatment of a 1, 4-diketone with ammonia. Propose amechanism. R + H20 RCCH2CH2R" R- NH3 R.
-
Suppose Nordic Boots was making production decisions for the following six boots: ot Price E(Demand) Std. Dev (Demand) Co Cu Exp. Profit Max. Profit A 220 1000 400 35.2 52.8 1101 39201 52800 230 2000...
-
What is parallel construction, and why is it important?
-
Find the piecewise-linear equivalent circuit for the diode of Fig. 1.15. Use a straight-line segment that intersects the horizontal axis at 0.7 V and best approximates the curve for the region...
-
The PFD in Figure 11.58 shows a process for the manufacture of \(\mathrm{X}\) and \(\mathrm{Y}\). The process feed is heated in furnace \(\mathrm{F}-100\) to \(800^{\circ} \mathrm{C}\) and is then...
-
A Venture Capitalist presents Arbuckle, Inc., the shoes manufacturer with the following term sheet for a series A funding round: Amount $5 million Security Convertible Preferred Mandatory on IPO >...
-
You are the budgeting manager for Summertime Grills Limited. The company makes two types of gas grills - the Tailgater and the Master Chef. For each of the two models, three raw materials are...
-
This comprehensive review problem requires you to complete the accounting cycle for Sherman Realty twice. This practice set allows you to review Chapters 1-5 while reinforcing the relationships...
-
Which of the following amino acids are more likely to be found on the outside of a globular protein, and which on the inside? Explain. (a) Valine (b) Aspartic acid (c) Phenylalanine (d) Lysine
-
An Fmoc protecting group can be removed from an amino acid by treatment with the amine base piperidine. Propose amechanism. pKg = 23 !! CH2 -NHCHCO- HoiHdo- NaOH H3NCHCO- Co2 2 Fmoc-protected amino...
-
Bernard Windows is a small company that installs windows. Its cost structure is as follows: Use (a) the equation method and (b) the contribution method to calculate operating income if Bernard...
-
address the following: What are the main considerations for program, portfolio, and project management? Discuss whether procurement can be eliminated or replaced by alternate organizational...
-
After you have read Beccaria and Lombroso, please provide a brief summary on these two theories of crime. Are these classical theories still relevant today? What are some of the criticism around...
-
Camille also wants to summarize the number of discounted products by product type and calculate their total and average discounted prices. In cell K2, enter a formula using the COUNTIF function that...
-
Managing project expectations is a central role for the project manager. How do bottlenecks limit this functionality and how can they be addressed? Under what circumstances would a project need to be...
-
23. Sally has hidden her brother's birthday present somewhere in the backyard. When writing instructions for finding the present, she used a coordinate system with each unit on the grid representing...
-
What equal monthly investment is required over a 40-year period to achieve a balance of $2,000,000 in an investment account that pays monthly interest of 3/4 percent?
-
In Exercises discuss the continuity of each function. f(x) -3 1 x - 4 y 3 2 -1 -2 -3+ 3 X
-
Determine whether each cation is acidic or pH-neutral. For those cations that are acidic, write an equation that shows how the cation acts as an acid. a. NHA b. Nat c. CO+ d. CHNH3
-
Write out a scheme similar to eq. 11.19 to show how you could separate a mixture of p toluidine, p-methylphenol, and p-xylene. CH3 NHz CH3 OH CH3 CH3 p-toluidine p-methylphenol p-xylene
-
Draw the important contributors to the resonance hybrid structure of p-nitroaniline (page 338).
-
Place the following substances, which have nearly identical formula weights, in order of increasing boiling point: CH3CH2CH2CH2NH2, CH3CH2CH2CH2OH, CH3CH2CH2OCH3, and CH3CH2CH2CH2CH3.
-
The market price of a semi-annual pay bond is $957.95. It has 15.00 years to maturity and a coupon rate of 7.00%. Par value is $1,000. What is the yield to maturity? The market price of a semi-annual...
-
Compute the accumulated sum of the following payments: (a) $400 every six months for 4 years at 8% compounded semi-annually (b) $250 per month for 5 years at 7.5% compounded monthly.
-
The output is wrong. Sometimes a program lacking input will produce wrong output (as in this case), or no output. Remember to always pre-enter needed input. Type 2 in the input box, then click "Run...

Study smarter with the SolutionInn App


