Consider the following two compounds: a) Identify which of these two compounds has greater resonance stabilization. b)
Question:
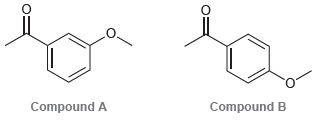
a) Identify which of these two compounds has greater resonance stabilization.
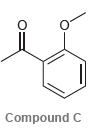
b) Would you expect compound C (below) to have a resonance stabilization that is more similar to compound A or to compound B?
Transcribed Image Text:
Compound B Compound A
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 75% (12 reviews)
a Compound B has one additional resonance structure that Compound A lacks becaus...View the full answer

Answered By

Kulwant Singh
I teaching mathematics from last 12 years to college students in government college in Punjab India.
I have also teaching as tutor to school students in my home. I have great interest in mathematics so I have pursuing phd in mathematics from Dravidian university. I tried my best to satisfie the students doubt.
Thanks
With regards
Kulwant Singh
0.00
0 Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
Identify the relationship between the following two compounds.
-
Consider the following two compounds. These compounds are stereoisomers of 1,2,3-trimethylcyclohexane. One of these compounds has three chirality centers, while the other compound has only two...
-
Consider the following two compounds. When treated with NaOH, one of these compounds forms an epoxide quite rapidly, while the other forms an epoxide very slowly. Identify which compound reacts more...
-
For each of the trees in the previous exercise, perform a find with path compression on the deepest node.
-
Using the Feynman rules for a \(\phi \bar{\psi} \psi\) Yukawa theory, write down the one-loop contribution to the \(\phi^{4}\) proper vertex. Show that this one-loop contribution is divergent and...
-
Which invention attributed to van Leeuwenhoek had a pronounced influence on the creation of the sciences of cytology, bacteriology, and pathology?
-
Water at 290 K and 0.2 kg/s flows through a Teflon tube (k = 0.35 W/m K) of inner and outer radii equal to 10 and 13 nun, respectively. A thin electrical heating tape wrapped around the outer...
-
Suppose, you want to start a small business. prepare at least 5 slides on what is your small business and outline some of the requirements to create and maintain it. What are few strategies to become...
-
Suppose we generate a training set from a decision tree and then apply decision-tree learning to that training set. Is it the case that the learning algorithm will eventually return the correct tree...
-
In the previous problem, we saw that an acetylide ion can attack a variety of electrophiles. In Chapter 20, we will see that a C=O bond can also function as an electrophile. Consider the following...
-
In each case below, identify the acid and the base. Then draw the curved arrows showing a proton transfer reaction. Draw the products of that proton transfer, and then predict the position of...
-
a. A city is considering airport expansion. In particular, the mayor and city council are trying to decide whether to sell bonds to construct a new terminal. The problem is that at present demand for...
-
1. Suppose the expected market risk premium is 7.5 percent and the risk free rate is 3.7 percent. The expected return on TriStar's stock is 14.2 percent. Assume the capital-asset-pricing-model holds....
-
A. Given below is information about three RM $5000 par value bonds, each of which pays coupon semiannually. The required rate of return on each bond is 12%. Calculate the value of the bonds and...
-
The following table for 5.5% par-value bonds with annual coupons will be used in this question: Term 1 year 2 year Yield 1.7% 2.45% 2.75% 3 year Use the above table to determine the 3 year spot rate....
-
1. A firm issues bonds with a maturity of 12 years, a 9% coupon rate, and a face value of $1,000. The bonds make annual coupon payments. If the yield to maturity is 11.5%, I what is the price of the...
-
How does the organization's board of directors demonstrate its commitment to effective internal control and risk management processes?
-
Fill in the blank with an appropriate word, phrase, or symbol(s). In the Borda ________________ voting method, the candidates are ranked from most favored to least favored.
-
State whether each statement is true or false. If false, give a reason. {purple, green, yellow} = {green, pink, yellow}
-
Among the most common over-the-counter drugs you might find in a medicine cabinet are mild pain relievers such ibuprofen (Advil, Motrin), naproxen (Aleve), and acetaminophen (Tylenol). (a) How many...
-
Which element in each of the following pairs is more electronegative? (a) Li or H (b) B or Br (c) C1 or I (d) C or h
-
Use the + / convention to show the direction of expected polarity for each of the following bonds indicated. (a) H3C C1 (b) H3C NH2 (c) H2N H (d) H3C SH (e) H3C MgBr (f) H3C F
-
Identify one type of business and describe that business and it's lending needs. Then describe what their potential lending needs may? Manufacturing Retail Wholesale Service Construction
-
Many investors believe the slope of yield curve is a leading economic indicator. Do you agree? Why? https://home.treasury.gov/policy-issues/financing-the-government/interest-rate-statistics?
-
Analyze the process for monitoring claims data for detecting potential fraud and abuse and describe how it is a crucial financial management process?

Study smarter with the SolutionInn App


