Consider the structure of formaldehyde: a) Identify the type of bonds that form the C = O
Question:
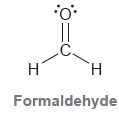
a) Identify the type of bonds that form the C = O double bond.
b) Identify the atomic orbitals that form each C = H bond.
c) What type of atomic orbitals do the lone pairs occupy?
Transcribed Image Text:
.C. H. Formaldehyde
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 62% (8 reviews)
a The CO bond of formaldehyde is comprised of one sigma b...View the full answer

Answered By

Ann Wangechi
hey, there, paying attention to detail is one of my strong points, i do my very best combined with passion. i enjoy researching since the net is one of my favorite places to be and to learn. i am a proficient and versatile blog, article academic and research writing i possess excellent English writing skills, great proof-reading. i am a good communicator and always provide feedback in real time. i'm experienced in the writing field, competent in computing, essays, accounting and research work and also as a Database and Systems Administrator
4.90+
151+ Reviews
291+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
Consider the structure of the catnip ingredient nepetalactone (page 305). a. Show with dotted lines that the structure is composed of two isoprene units. b. Circle the stereogenic centers and...
-
Consider the structure of prostaglandin E2 shown on page 451. a. How many stereogenic centers are present? b. What is the configuration (R or S) of each? c. What is the configuration of the double...
-
Consider the structure of the following compound: (a) When this compound is treated with bromine under conditions that favor monobromination, two stereoisomeric products are obtained. Draw them, and...
-
The input file for this assignment is Weekly_Gas_Average.txt. The file contains the average gas price for each week of the year. Write a program that reads the gas prices from the file into an...
-
How is a bank a manufacturing business? (Think of a bank's "production function.")
-
We define the even and odd parts of a sequence \(x(n), \mathcal{E}\{x(n)\}\) and \(\mathcal{O}\{x(n)\}\) respectively, as \[\begin{aligned}\mathcal{E}\{x(n)\} & =\frac{x(n)+x(-n)}{2}...
-
Match each five number summary with one of the histograms in Figure 2.23. The scale is the same on all four histograms. (a) $(1,3,5,7,9)$ (b) $(1,4,5,6,9)$ (c) $(1,5,7,8,9)$ (d) $(1,1,2,4,9)$ Figure...
-
A company must decide whether to provide their salesmen with company-owned automobiles or to pay the salesmen a mileage allowance and have them drive their own automobiles. New automobiles would cost...
-
What role does emotional intelligence play in conflict management, and how can emotional regulation techniques enhance conflict resolution effectiveness in interpersonal and organizational contexts ?
-
Write a Super class interface employee has name and id number. Write manager and labour derived from employee class. Manager class has member data qfunction and qualification and manager allowance...
-
In each of the following transformations, identify whether the starting material has been oxidized, reduced, or neither. Try to determine the answer without calculating Oxidation states, and then use...
-
Identify which two compounds below are constitutional isomers: (CH].C,H, (CH),c, (CH),,
-
Use the solubility product constant from Appendix F to determine whether a precipitate will form if 20.0 mL of 1.0 10 -6 M magnesium chloride is added to 80.0 mL of 1.0 10 -6 M potassium fluoride....
-
SportZ has invoices for materials purchased from Platinum Steel Incorporated. The invoices are for $4,242 due 60 days ago, $12,567 due in 30 days, and $18,451 due in 140 days. If SportZ pays all of...
-
Kaiyu has a $40,000 car loan at 12% for 36 months, on which she makes monthly payments of $1,328.57. After making her 10th payment, she wants to know the amount to pay the loan off. What is the...
-
1 A soup recipe calls for 3 cups of chicken broth and will make enough to serve 10 people. How much broth is required if the recipe is modified to serve 6 people? The recipe will require cups of...
-
The following scatterplot is created using one of the datasets in the income and housing data in the spreadsheet linked above. 40 35 30 25 20 20 15 10 5 0 500 550 600 650 Which variables are used in...
-
7. In the coin-flipping situation of problem 1, what is P(H = 4| H is even)?
-
The following table gives the distribution of annual salaries for employees at Kulzers Home Improvement. Annual Salary . . . . . . . . . . . . . . . . Number Receiving Salary $100,000 . . . . . . . ....
-
President Lee Coone has asked you to continue planning for an integrated corporate NDAS network. Ultimately, this network will link all the offices with the Tampa head office and become the...
-
Name and assign R or S stereochemistry to the product(s) you would obtain by reaction of the following substance with ethyl magnesium bromide. Is the product chiral? Is it optically active? Explain.
-
Give IUPAC names for the following compounds: (a) (b) (c) CH CHCHCH2CH CH2CH2CH3 H-CH2CHCH20H (d) (e) Ph. (f) Br NEC-
-
Draw and name the eight isomeric alcohols with formula C5H12O.
-
Solve (2.4 x 10-5) X 875 (2.5 x 107) x (2.8 x 107)
-
assume there are no outliers. Draw the boxplot for the following frequency table. You may X f rf cf crf 3 7 0.1228 7 0.1228 4 11 0.1930 18 0.3158 5 9 0.1579 27 0.4737 618 6 0.1053 33 0.5789 10 0.1754...
-
Find the partial derivative of the regularized least squares problem: {) (w + x) (wo + w x ( ) + w x ( ) } + |/\||[w1, w2]|| with respect to wo, w, and w2. Although there is a closed-form solution to...

Study smarter with the SolutionInn App


