Consider the structure of N,N-dimethylformamide (DMF): We might expect the two methyl groups to be equivalent; however,
Question:
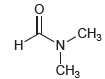
We might expect the two methyl groups to be equivalent; however, both the proton and carbon NMR spectra of DMF show two separate signals for the methyl groups. Propose an explanation for the nonequivalence of the methyl groups. Would you expect the signals to collapse into one signal at high temperature?
Transcribed Image Text:
CHз N. Н CHз
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 100% (6 reviews)
NNdimethylformamide DMF has several resonance structures Consider the thi...View the full answer

Answered By

Somshukla Chakraborty
I have a teaching experience of more than 4 years by now in diverse subjects like History,Geography,Political Science,Sociology,Business Enterprise,Economics,Environmental Management etc.I teach students from classes 9-12 and undergraduate students.I boards I handle are IB,IGCSE, state boards,ICSE, CBSE.I am passionate about teaching.Full satisfaction of the students is my main goal.
I have completed my graduation and master's in history from Jadavpur University Kolkata,India in 2012 and I have completed my B.Ed from the same University in 2013. I have taught in a reputed school of Kolkata (subjects-History,Geography,Civics,Political Science) from 2014-2016.I worked as a guest lecturer of history in a college of Kolkata for 2 years teaching students of 1st ,2nd and 3rd year. I taught Ancient and Modern Indian history there.I have taught in another school in Mohali,Punjab teaching students from classes 9-12.Presently I am working as an online tutor with concept tutors,Bangalore,India(Carve Niche Pvt.Ltd.) for the last 1year and also have been appointed as an online history tutor by Course Hero(California,U.S) and Vidyalai.com(Chennai,India).
4.00+
2+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
Consider the structure of the catnip ingredient nepetalactone (page 305). a. Show with dotted lines that the structure is composed of two isoprene units. b. Circle the stereogenic centers and...
-
Consider the structure of prostaglandin E2 shown on page 451. a. How many stereogenic centers are present? b. What is the configuration (R or S) of each? c. What is the configuration of the double...
-
Consider the structure of the following compound: (a) When this compound is treated with bromine under conditions that favor monobromination, two stereoisomeric products are obtained. Draw them, and...
-
develop a trial balance, complete with an appropriate three-line heading 1. Bought office equipment from Peckoff Equipment Co. on 30-day credit terms, $1,390. Purchase Invoice #2071. Ledger Entries:...
-
What are three examples of code of ethics outlined by the Association for Computing Machinery (ACM)?
-
Using statistical software or a graphing calculator, determine the five-number summary of the data presented in Table 17. Approach We will use Minitab to obtain the five-number summary. Table 17...
-
Refer to Exercise 4.80 and obtain the (a) mean; (b) variance; (c) standard deviation for the number of requests for conference rooms. Data From Exercise 4.80 4.80 Upon reviewing recent use of...
-
Justin Fawber is a divisional manager within Monmouth GPS Guidance Systems. Haas is concerned about the amount of the divisions production. The following production data are available for April 2013:...
-
How did they calculate the Max debt and Min debt as well as the WACC? According to the rating category, if Deluxe tends to maintain at AA rating. The Long-term debt/capital (%) should between -1.1%...
-
Here are incomplete financial statements for Liam, Inc. InstructionsCalculate the missing amounts. LIAM, INC. Balance Sheet Liabilities and Stockholders' Equity Assets $ 7,000 10,000 45,000 Cash...
-
Deduce the structure of a compound with molecular formula C 5 H 10 O that exhibits the following IR, 1 H NMR, and 13 C NMR spectra. Data from the mass spectrum are also provided. 100 Mass Spec. Data...
-
Consider the structure of phenol: The chemical shift of the hydroxyl proton is found to be sensitive to the concentration of phenol. In a concentrated solution, the hydroxyl proton produces a signal...
-
Abbott Laboratories makes and sells Ensure, a nutritional supplement beverage (NSB). Ensure has consistently held the greatest market share of all NSBs on the market. Gerber Products Co. reformulated...
-
View Policies Current Attempt in Progress Sunland Enterprises purchased equipment on March 15, 2024, for $76,110. The company also paid the following amounts: $540 for freight charges; $196 for...
-
Hudson Community College enrolls students in two departments, Liberal Arts and Sciences. The college also has two service departments, the Library and the Computing Services Department. The usage of...
-
Bal., 1,600 units, 35% completed 17,440 To Finished Goods, 29,600 units ? Direct materials, 29,000 units @ 275,500 $9.50 Direct labor Factory overhead 84,600 39,258 Bal. ? units, 45% completed...
-
2 Bergo Bay's accounting system generated the following account balances on December 31. The company's manager knows something is wrong with this list of balances because it does not show any balance...
-
How did the radical Anabaptist and the protestant reformers, Luther, Calvin, and Zwingli differed in their theological views in relation to Scripture?
-
The following information was drawn from the annual report of Machine Imports Company (MIC): Required a. Compute the percentage of growth in net income from Year 1 to Year 2. Can stockholders expect...
-
A consultant is beginning work on three projects. The expected profits from these projects are $50,000, $72,000, and $40,000. The associated standard deviations are $10,000, $12,000, and $9,000....
-
Rank the following sets of substituents in order of priority according to the Cahn-lngold?Prelog sequence rules: (a) -CH, -Br, -,-1 (b) -, -, -, -2 (c) -, -2CH3, -, - (d) -CH, -H2H, -H2H2, -H (e)...
-
An Assign E or Z configuration to each of the following alkenes: ( - C (a) H2 C=C C=C H3C ci OCH3 (c) (d) CH3O2C CH=CH2 NC CH C=C CH3CH2 CH2 HO2C CH-CH
-
Name the following cycloalkenes: (c) (b) (a) CH (f) (e) (d)
-
Write an equation for the polynomial graphed below 5+ 4 + -5-4-3-2 y = 3. 2- 1 + + 2 3 4 5 -2 345 -5+
-
How can ethical leadership principles be leveraged to navigate the complexities of organizational dynamics and promote a culture of integrity, accountability, and social responsibility ?
-
Consider the 12-QAM constellation shown in Figure 2 and the baseband pulse, g(t) depicted in Figure 3. The 12 passband transmit waveforms are represented as Sm1(t) = Amig(t) cos (2fet) - Amag(t) sin...

Study smarter with the SolutionInn App


