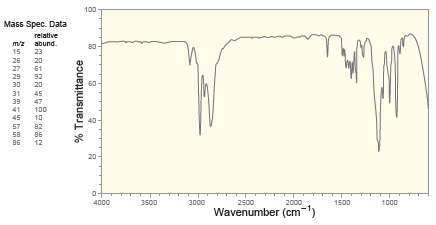
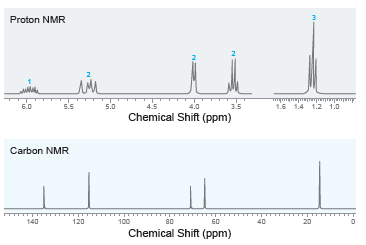
Deduce the structure of a compound with molecular formula C 5 H 10 O that exhibits the
Question:
Transcribed Image Text:
100 Mass Spec. Data relative mz abund. 15 23 26 27 29 20 61 92 30 20 31 39 47 41 100 45 10 57 82 58 86 86 12 20 4000 3500 3000 2500 2000 1500 1000 Wavenumber (cm-") % Transmittance Proton NMR 1.5 14 1.2 1.0 6.0 5.5 5.0 4.5 4.0 3.5 Chemical Shift (ppm) Carbon NMR 140 120 100 80 60 20 Chemical Shift (ppm)
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 72% (18 reviews)

Answered By

Dinesh F
I have over 3 years of professional experience as an assignment tutor, and 1 year as a tutor trainee.
5.00+
1+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
Deduce the structure of a compound with molecular formula C 9 H 10 O 2 that produces the following 1 H NMR spectrum and 13 C NMR spectrum: Proton NMR 10 Chemical Shift (ppm) Carbon NMR - 128.4 128.8-...
-
The IR spectrum of a compound with molecular formula C5H8O was obtained in CCl4 and is shown in Figure 13.42. Identify the compound. Wavelenga qum) 15 16 14 3600 340) 3800 3300 3000 280K 2600 2400...
-
The 1 H NMR spectrum of a compound with molecular formula C 7 H 15 C l exhibits two signals with relative integration 2 : 3. Propose a structure for this compound.
-
You work for North Carolina Parks and Recreation. Do you support or oppose the idea to unionize federal and state employees? Explain your thoughts below.
-
What are two types of information available on the Web? Which type can be censored?
-
Use the results from Example 1 to construct a boxplot of the finishing times of the men in the 60- to 64-year-old age group. Data from Example 1 The data shown in Table 17 show the finishing times...
-
Determine whether the following can be probability distributions of a random variable that can take on only the values of 0,1 , and 2 : (a) \(f(0)=0.34 \quad f(1)=0.34\) and \(f(2)=0.34\). (b)...
-
Prepare a tax return for Charlie and Cindy Clergy. Charlie Clergy (age 44, DOB: 5/2/76, SSN: 453-00-9863), whose Form W-2s and 1099s follow, is an ordained minister in the Episcopal Church. His wife,...
-
What is the output of the following C code? 1 #include 2 3 void xMethod(int n) { 5 } 676945 8 10 11 printf("%d",n+1); void xMethod(double n) { printf("%f",n+2); } void xMethod(long n) { 12 13. } 14...
-
A ride hailing company has their DB structured in 3 major tables as described in the SCHEMA section below. Write a query to fetch the top 100 users who traveled the most distance using the service....
-
For each of the following compounds, determine whether the two protons shown in red are homotopic, enantiotopic, or diastereotopic: (a) (b) (c) (d) (e) Discuss. OMe . CI H,
-
Consider the structure of N,N-dimethylformamide (DMF): We might expect the two methyl groups to be equivalent; however, both the proton and carbon NMR spectra of DMF show two separate signals for the...
-
In 2012, 14.8% of medium-sized businesses in Canada were foreign-controlled, according to Statistics Canada Table 1790005: Corporations Returns Act (CRA), enterprise characteristics by country of...
-
On 31 December 2020, the total account receivable for Beauty Enterprise is RM30,000, 5% of these debts are estimated to go bad in the foreseeable future. Required: iv. Calculate Allowance for...
-
If the total cost of producing 20 units of output is $1000 and the average variable cost is $35, what is the firm's average fixed cost at that level of output?
-
In December 2019, an infectious disease caused by severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2) was first reported in Wuhan City, Hubei Province of China which was later called Corona...
-
Prepare budgetary entries, using general ledger control accounts only, for each of the following unrelated situations: (If no entry is required for a transaction/event, select "No Journal Entry...
-
What assumption is NFPA 101 built upon with regards to the number of fire sources? Why might this be inadequate in the case of incendiary or set fires?
-
Sam and his sister Blair both attend the state university. As a reward for their successful completion of the past year (Sam had a 3.2 GPA in business, and Blair had a 3.7 GPA in art), their father...
-
As long as we can't lose any money, we have a risk-free investment." Discuss this comment. Q2: Both investing and gambling can be defined as "undertaking risk in order to earn a profit." Explain how...
-
The heat of hydrogenation for allene (Problem 6.37) to yield propane is 295kJ/mol, and the heat of hydrogenation for a typical monosubstituted alkene such as propene is 126kJ/mol. Is allene more...
-
Predict the major product in each of the following reactions: CH (a) H20 CHCH-CH3H2CH H2SO4 (Addition of H20 occurs.) (b) CH-CH CH (c) HBr (d) 2 HCI %3 CHCH2CH2CH2CH%3DCH2
-
Predict the major product from addition of HBr to each of the following alkenes: (a) CH2 (b) (c) CH CH3CHHHH
-
Paraphrase this "To fight the 2008 financial crisis, the United States employed a combination of fiscal and monetary policies. On the budgetary front, Congress passed the ailing Asset Relief Program...
-
a) A company just paid a dividend of Do = $5.00 on its preferred stock. The dividend amount will remain constant. The discount rate (i.e., market capitalization rate) of the company is k = 8%. What...
-
Jessica is planning to paint her room. It measures 11 feet by 13 feet and has 8 -foot ceilings. One long wall and one short wall each have a window that measures 3 feet by 4 feet. One short wall has...

Study smarter with the SolutionInn App


