Question: Consider the two methyl groups shown in the following compound. Explain why the methyl group on the right side appears at lower chemical shift. CH3
Consider the two methyl groups shown in the following compound. Explain why the methyl group on the right side appears at lower chemical shift.
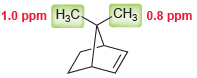
CH3 0.8 ppm 1.0 ppm H;C.
Step by Step Solution
3.50 Rating (163 Votes )
There are 3 Steps involved in it
The methyl group on the righ... View full answer

Get step-by-step solutions from verified subject matter experts


