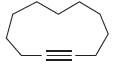
Draw the major product(s) expected when each of the following alkynes is treated with aqueous acid in
Question:
(a)
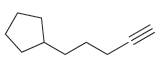
(b)
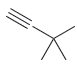
(c)
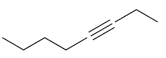
(d)
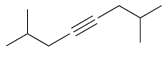
(e)
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 66% (9 reviews)
a...View the full answer

Answered By

Munibah Munir
I've done MS specialization in finance’s have command on accounting and financial management. Forecasting and Financial Statement Analysis is basic field of my specialization. On many firms I have done real base projects in financial management field special forecasting. I have served more than 500 Clients for more than 800 business projects, and I have got a very high repute in providing highly professional and quality services.I have capability of performing extra-ordinarily well in limited time and at reasonable fee. My clients are guaranteed full satisfaction and I make things easy for them. I am capable of handling complex issues in the mentioned areas and never let my clients down.
4.60+
467+ Reviews
648+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
Draw the major product(s) that are expected when each of the following amines is treated with excess methyl iodide and then heated in the presence of aqueous silver oxide. (a) (b) NH2 NH2
-
Draw the condensation product that is expected when each of the following esters is treated with sodium ethoxide followed by acid workup. (a) (b) (c) OEt OEt
-
Draw the major product expected when 1,3-butadiene is treated with one equivalent of HBr at 0C, and show the mechanism of its formation.
-
Find the dy/dx for the following 3 2x+1.2x+1 (x +1)4 1- y=-
-
John Clinton, owner of Clinton Company, applied for a bank loan and was informed by the banker that audited financial statements of the business h ad to be submitted before the bank could consider...
-
The basal transcription apparatus is composed of a. five general transcription factors. b. RNA polymerase II. c. a DNA sequence containing a TATA box and transcriptional start site. d. all of the...
-
With Figure 1.5 in mind, how could changes in government policies affect accounting activities? All Stakeholders Financial statements Bank & agency reports Accounting System Tax returns Management &...
-
Thomson's Model of the Atom In the early years of the 20th century, a leading model of the structure of the atom was that of the English physicist J. J. Thomson (the discoverer of the electron). In...
-
Marin Company produces two software products ( Cloud - X and Cloud - Y ) in two separate departments ( A and B ) . These products are highly regarded network maintenance programs. Cloud - X is used...
-
Twenty percent of Americans ages 25 to 74 have high blood pressure. If 16 randomly selected Americans ages 25 to 74 are selected, find each probability. a. None will have high blood pressure. b....
-
Consider the following equilibrium: These constitutional isomers rapidly interconvert in the presence of even trace amounts of acid and are therefore said to be tautomers of each other. Draw a...
-
What are three U.S. ecosystems that need protection?
-
The Kool King Company has followed a policy of no layoffs for most of the manufactures tile, even though the demand for its air conditioners is highly seasonal. Management wants to evaluate the cost...
-
An investor company owns 30% of the outstanding common stock of an investee company, which allows the investor to exercise significant influence over the investee. The Equity Investment was reported...
-
Worker's Compensation provides the employee with protection but also protects the employer from what? Explain.
-
US Bank has US$200 million worth of one year loans earing an average rate of Return of 4%. The bank has one year single payment Canadian dollar loans of C$110 million earning 6%. The banks funding...
-
Voice and Accountability captures perceptions of the extent to which a country's citizens are able to participate in selecting their government, as well as freedom of expression, freedom of...
-
Who is generally responsible for providing application specifications to the Application Developer?
-
Kwame borrowed $2500 for 5 months from a bank using a 3% discount note. (a) How much interest did Kwame pay the bank for the use of its money? (b) How much did he receive from the bank? (c) What was...
-
Synthesize the products by drawing out reagents and intermediates along the way. `N H. OH HO HO
-
In light of your answer to Problem 11.49, which alkene, F or Z, would you expect from an E2 reaction on the tosylate of (2R, 3R)-3-phenyl-2-butanol? Which alkene would result from E2 reaction on the...
-
How can you explain the fact that franc-i -hromo-2-methylcyclohcxane yields the non-Zaitsev?s elimination product 3-methylcyclohexene on treatment with base? H C Br trans-1-Bromo-2-methylcyclohexane...
-
Predict the product(s) of the following reaction, indicating stereochemistry wherenecessary: Br CH3 H20 Ethanol
-
Jen and Barry's ice cream shop charges $1.65 for a cone. Variable expenses are $0.31 per cone, and fixed costs total $2,000 per month. A Valentine's Day promotion is being planned for the second week...
-
a. The future value of a $1,050 savings deposit after five years at an annual interest rate of 4 percent. (Round FV factor to 3 decimal places and final answer to 2 decimal places.) Future value b....
-
QuestromT Not complete Marked out of 2.00 ring question Homework - Week 6 - Module 23 Determining Unit Costs, Variance Analysis, and Interpretation Big Dog Company, a manufacturer of dog food,...

Study smarter with the SolutionInn App


