Predict the products that are expected when each of the following alkenes is treated with a peroxy
Question:
a.
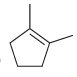
b.
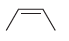
c.
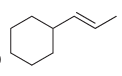
d.
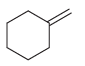
e.
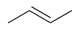
f.
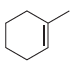
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 50% (8 reviews)
a b ...View the full answer

Answered By

MICHAEL K L
Let explore the education and i am having the experience. I have done the graduation in 2013
0.00
0 Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
Predict the products that are expected when each of the following compounds is heated with concentrated HBr. a. b. c. d.
-
Draw the products that are expected when each of the following amino acids is treated with ninhydrin: (a) l-Aspartic acid (b) l-Leucine (c) l-Phenylalanine (d) l-Proline
-
When each of the following ketones is treated with aqueous sodium hydroxide, the aldol product is obtained in poor yields. In these cases, special distillation techniques are used to increase the...
-
A small company has $4,500,000 in (annual) revenue, spends 57% of its revenues on purchases, and has a net profit margin of 11.75%. They would like to increase their profits and they are looking at...
-
A current problem in the U.S. Army is the neck/shoulder fatigue experienced by helicopter pilots. To be able to fly missions at night, the pilots wear night vision goggles, which are attached to the...
-
Naihui and Haiyan deposit \($250\) into their joint account at the end of each month. They want to have a total of \($12\),000 in their account after 40 months. a. What monthly rate of interest must...
-
A sage of selling once said, Your job as a salesperson is to do 80 percent listening and 20 percent talking. Do you agree? Why or why not?
-
Heavenly Displays Inc. puts together large-scale fireworks displaysprimarily for Canada Day celebrations sponsored by corporations and municipalities. The company assembles and orchestrates complex...
-
M&R Company provided $ 2 , 0 0 0 in services to customers in December, which are not yet recorded. Those customers are expected to pay the company in January following the company s year - end. Wage...
-
What is the difference between subsequent revenue expenditure and a subsequent capital expenditure, and how should they be recorded?
-
The following reaction is very slow. Identify the mechanism, and explain why the reaction is so slow. Br NaOH,
-
For each of the products shown in the following reaction, propose a mechanism that explains its formation: Br NBS, hv Br
-
Write at least three examples of hypotheses that involve a managerial action statement. Provide a corresponding decision statement for each.
-
Consider an Sn2 reaction of NaSH in a polar aprotic solvent with (CH3)2CHCH2CH2X. What would be the relative order of reactivity for the following X substituents? I. X= I II. X=Br III. X=Cl IV. X=F...
-
Tom sold mutual fund share he had owned 3 years so that he could use the precedes to return to college. Tom is in the 15% marginal tax bracket and his capital gains from this sale were $64,300. How...
-
A car costs $27,000 in the U.S. The same car costs 32,130 Euros. Based on these data points, please answer the below (2 decimal places). (show work along with answer) What should EUR/USD be in real...
-
Cash Flow. College students often have little income and many expenses. Does this reduce or increase the importance of completing a cash-flow statement on a monthly basis? Why?
-
The Government of Canada Zero Coupon Yield curve is shown below. What does the shape over the first three years tell you about the market's consensus expectation for future spot interest rates?...
-
After toxic fumes leaked from Union Carbides pesticide plant in Bhopal, India, more than 3,300 people were killed and another 30,000 injured. Two sources later indicated that Union Carbide had...
-
President Lee Coone has asked you to continue planning for an integrated corporate NDAS network. Ultimately, this network will link all the offices with the Tampa head office and become the...
-
Show the hybridization at each of the atoms, except H, in these molecules. Indicate the type of each designated bond and the orbital's that are overlapping to form it? (both) TTT a) HC=C_C_CH ...
-
What is the hybridization at all atoms, except hydrogen's, in these compounds? a) H H H H H H H H CH 4.6 c) HC-N-CH3 : : g) CHC-OH h) CHC-NHCH,
-
Draw the p orbital's that compose the conjugated part of these molecules: a) :-CH3 b) CH,=CHNH, c) H-C-C-CH=CH
-
July August September October November December Total Average 350 450 460 390 390 360 4,960 413 3,020 3,500 4,020 3,290 3,170 3,070 $ 43,571 $ 3,631 PR 6-38 (Algo) Part 4 Compute the variable cost...
-
Discuss the main distinguishing features of majority, concurring, and dissenting opinions
-
On September 3, 2023, Wilkins purchased 1244 stock in Brass Corporation for $6,000. On December 31, 2023, the stock was worth $8,500. On August 15, 2024, Wilkins was notified that a. 2023-$0;...

Study smarter with the SolutionInn App


