For each of the following pairs of alcohols, identify the one that is more acidic, and explain
Question:
a.
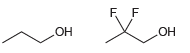
b.
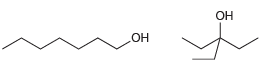
c.
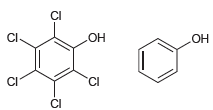
d.
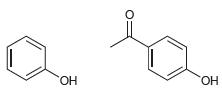
e.
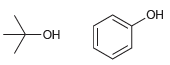
Transcribed Image Text:
но но F. OH
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 77% (9 reviews)
a The electronwithdrawing effects of the fluorine atoms stabilize the conjugate base b The conjug...View the full answer

Answered By

Saleem Abbas
Have worked in academic writing for an a years as my part-time job.
5.00+
1+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
For each of the following pairs of compounds, give one absorption band that could be used to distinguish between them: a. b. c. d. e. f. CH3CH2CH2CH and CH3CH2OCH3 CH CH2COCH and CH3CH2COH CH3CH2COH...
-
For each of the following pairs of compounds, identify one IR absorption band that could be used to distinguish between them: a. b. c. d. e. f. g. h. i. cis-2-butene and trans-2-butene j. CH3CH2CH2OH...
-
For each of the following pairs of compounds, indicate the compound that you would expect to be a more potent inhibitor of dihydrofolate reductase: a. b. NH NH CI of CH3 CH3 NH2 CH3 or H.NNCHCHCHCH,...
-
1. Explain the Theories of leadership 2. Research -In modern times give 1 example as to which business leader in the hospitality industry has made a mark and why.?
-
Write a program to evaluate a postfix expression.
-
The after-tax wealth in Kaminskis tax-deferred account at the end of the five years will be closest to: A. \($1\),122,041. B. \($1\),313,166. C. \($1\),402,552. Jevan Chen is a tax adviser who...
-
Mr. A. Gaylord manages a pension fund and believes that his stock selection ability is excellent. However, he is worried because the market could go down. He considers entering an equity swap where...
-
A Wall Street Journal article reported that 15 of the 30 NBA teams have employees whose sole responsibility is to analyze data. The information compiled by these statheads is then used to help make...
-
The rate of discount is given as d = (R-P)/R.n Where R is redemption value, P is initial price and n is time to redemption If the current rate of discount on treasury bills is quoted at 3%: i....
-
Enter the transactions for Weeks 1 5 into the appropriate Journal for the month of June 2022. Next, post all the Journal transactions to the ledger accounts as specified in the Account System...
-
The negative externality associated with self-protection from terrorism suggests that a. Terrorists cause more damage than they think they will. b. People engage in less self-protection than they...
-
Under many economic models of terrorism, the terrorist is assumed to act a. Without regard for incentives, costs, or benefits. b. In a predictable way, since they maximize costs subject to minimizing...
-
How do you define a view that displays only science-fiction books?
-
Montoure Company uses a perpetual inventory system. It entered into the following calendar - year purchases and sales transactions. Date Activities Units Acquired at Cost Units Sold at Retail January...
-
Statement of Retained Earnings Night Scapes, Corporation began the year 2 0 1 8 with $ 2 8 million in retained earnings. The firm suffered a net loss of $ 2 . 8 million in 2 0 0 8 and yet paid $ 2 ....
-
On May 1 , 2 0 2 5 , Sunland Company purchased factory equipment for $ 7 5 7 8 0 0 . The asset's useful life in hours is estimated to be 2 2 0 0 0 0 . The estimated salvage value is $ 3 6 0 0 0 and...
-
1. What does it mean for you to have a commitment to anti-racism? 2. How have you demonstrated that commitment and how would you see yourself demonstrating it at your workplace?
-
Crane Company purchased a depreciable asset for $ 1 2 4 0 0 0 0 . The estimated salvage value is $ 6 2 0 0 0 , and the estimated useful life is 1 0 0 0 0 hours. Crane used the asset for 1 1 0 0 hours...
-
Determine an Euler circuit that begins and ends with vertex B. Use the following graph. E.
-
Why is the national security argument for tariffs questionable?
-
Draw the product from reaction of each of the following substances with (i) Br2, FeBr3 and (ii) CH3COCl,AlCl3. (b) (a)
-
The following molecular model of a dimethyl-substituted biphenyl represents the lowest-energy conformation of the molecule. Why are the two benzene rings tilted at a 63 o angle to each other rather...
-
How would you synthesize the following compound starting from benzene? More than one step isneeded.
-
1. At the break-even point, Jefferson Company sells 95,000 units and has fixed cost of $351,500. The variable cost per unit is $0.30. What price does Jefferson charge per unit? Note: Round to the...
-
Your company wants you to research the potential for implementing a relational database. How would you explain a relational database to them? What are the pros and cons of relational databases? What...
-
In Exhibit 13-4, the makers of Healthy Hands Lotion discovered that the lotion can cause skin reactions, but it doesn't inform the buyers. While the market experiences imperfect information, the...

Study smarter with the SolutionInn App


