For each of the following substrates, determine whether an E1 process will require the use of an
Question:
a.
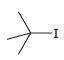
b.
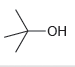
c.
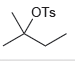
d.
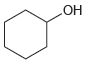
e.
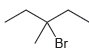
f.
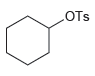
Transcribed Image Text:
-ОН
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 100% (5 reviews)
a No the leaving group is not OH b Yes ...View the full answer

Answered By

Akshada Ghangale
I have my tutoring experience on other tutoring platform named chegg
0.00
0 Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
For each of the following situations? Determine whether a long or short hedge is appropriate. Justify your answer. a. A firm anticipates issuing stock in three months. b. An investor plans to buy a...
-
For each of the following studies, say whether you would use a t test for dependent means or a t test for independent means. (a) A researcher randomly assigns a group of 25 unemployed workers to...
-
Determine the relaxation time for each of the following medium: (a) Hard rubber ( = 1015 S/m, = 3.1o) (b) Mica ( = 1015 S/m, = 6o) (c) Distilled water ( = 104 S/m, = 80 o)
-
Cadmium is a highly toxic substance with lethal effects on the human body in concentrations greater than 4.4 x 10-mol/L. Cadmium (II) carbonate has a Kp of 5.2 x 10-12 at 25C. Calculate the...
-
Based on, Kerzner, Harold. (2013) Project Management: A Systems Approach to Planning, Scheduling, and Controlling, 11th Ed. John Wiley & Sons. ISBN 978-1118022276.(Chapter 17) and Kerzner, Harold....
-
The data represent the number of driving fatalities for a certain area by age for male and female drivers. (a) What is the probability that a randomly selected driver fatality who was male was 16 to...
-
Frequently, sales managers use contests and recognition rewards to motivate the sales force. If sales managers understand salesperson performance, why is it necessary to employ these additional...
-
Calculate the following ratios for Lake of Egypt Marina, Inc. as of year-end 2018? Use the following financial statements for Lake of Egypt Marina, Inc. Calculate the following ratios for Lake of...
-
As of June 30, Year 1, the bank statement showed an ending balance of $16,878. The unadjusted Cash account balance was $15,239. The following information is available: 1. Deposit in transit, $2,190....
-
What is the solution of the recursive equation T (n) = 0.015625T +n?? (1/4) a) O(n') b) O(n' lg n) c) O(n* Ign) d) O(7*)
-
What are fossil fuels?
-
How are coal, oil, and natural gas formed?
-
Provide a two-or three-sentence response that argues for or against (indicate which) each of these statements: 1. Accounting is an exact science. 2. Managers choose accounting procedures that produce...
-
Let w = F(x, y), where x=u 3uv-v and y = g(u, v), F and g are differentiable, and g(5, 1) =0, gu (5, 1) = 29, 9, (5, 1) = 6, F(9, 0) 3, F(9, 0) = 14, Fy(9, 0) = 5. Iw Find when u 5 and v = 1. Type...
-
Steve has been operating Castle Creek Restaurant in Ontario for the past several years. On the basis of the information that Steve's accountant filed with the CRA during the prior year, Castle Creek...
-
Express the following comparative income statements in common-size percents. Using the common-size percents, which item is most responsible for the decline in net income? Complete this question by...
-
The following information is available for ACT230 Company: Beginning inventory First purchase 600 units at $4 900 units at $6 500 units at $7 Second purchase Assume that ACT 230 uses a periodic...
-
3 41 If AT-12 2 and B = [ 0 1 then find AT-BT -1 2 1 ], 1 2 3
-
Is the rate of cooling of a red-hot poker greater in a warm oven than in a cold room? (Or is the rate the same for each?)
-
(a) Use integration by parts to show that (b) If f and g are inverse functions and f' is continuous, prove that (c) In the case where f and t are positive functions and b > a > 0, draw a diagram to...
-
How strong a base would you expect a Grignard reagent to be? Look at Table 8.1, and then predict whether the following reactions will occur as written.?(The p K a of NH 3 is 35.) (a) CH 3 MgBr + H ?...
-
How might you replace a halogen substituent by a deuterium atom if you wanted to prepare a deuteratedcompound? Br , CHH-H CHCH2CH
-
How would you carry out the following transformations using an organo copper coupling reaction? More than one step is required in eachcase. (a) "CH (b) HH2CH2CHBr CH3CH2CH2CH2CH2CH2CH2CH3 (c)...
-
Lake Incorporated and the River, Incorporated reported net incomes of $205,000 and $164,000, respectively, for the most recent fiscal year. Both companies had 41,000 shares of common stock issued and...
-
What effect Eventually, BREXIT will make on EU? How it will benefit UK? What will be the possible outcome?
-
Merchant Company purchased property for a building site. The costs associated with the property were: Purchase price Real estate commissions Legal fees Expenses of clearing the land Expenses to...

Study smarter with the SolutionInn App


