Guanidine is a neutral compound but is an extremely powerful base. In fact, it is almost as
Question:
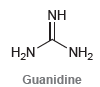
Transcribed Image Text:
NH H,N NH2 Guanidine
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 78% (14 reviews)
Protonation of the nitrogen highlighted below ...View the full answer

Answered By

Anoop V
I have five years of experience in teaching and I have National Eligibility in teaching (UGC-NET) .
5.00+
1+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
Histidine possesses a basic side chain which is protonated at physiological pH. Identify which nitrogen atom in the side chain is protonated.
-
Clomipramine is marketed under the trade name Anafranil and is used in the treatment of obsessive compulsive disorder. (a) Identify which nitrogen atom in clomipramine is more basic, and justify your...
-
Arginine is the most basic of the 20 naturally occurring amino acids. At physiological pH, the side chain of arginine is protonated. Identify which nitrogen atom in the side chain is protonated.
-
A bartender employed in a licensed establishment over-serves a patron. As a result of the over-service, the patron physically assaults another patron by striking him with a beer bottle. Identify and...
-
In 2015, there were approximately 8.3 million unemployed workers in the United States. The circle graph shows the age profile of these unemployed workers. (a) Estimate the number of unemployed...
-
There was a lengthy debate about whether to include a public option in health reform. A public option is some type of government-run health plan that would be available to compete with private plans....
-
Recall that simulated squared values from Exercise 2.7 follow F distributions. Use geom_qq and geom_qq_line to plot Q-Q plots of the simulated squared values against the appropriate $\mathrm{F}$...
-
An 11-m beam is subjected to a load, and the shear force follows the equation V(x) = 5 + 0.25x2 Where V is the shear force and x is length in distance along the beam. We know that V = dM/dx, and M is...
-
You take a long position in a one-year forward contract on a stock whose price you expect to increase in the future. The current price of the stock is $100 per share. The interest rate is 10%...
-
14. Isaiah's employer has a pension plan that pays 2,2% of the average of his highest three-year salary for every year worked, Isaiah has been with this company for 15 years, and his last three...
-
Propose an efficient synthesis for each of the following compounds using the acetoacetic ester synthesis. (a) (b) (c) (d)
-
Complete a situation analysis for Fork and Dagger, highlighting possible order qualifiers and order winners for the food and hospitality industry in Struan.
-
Your swimming pool is square and 5.0 m on a side. It is 3.0 m deep in the morning. If the temperature changes by 20C during the afternoon, how much does the depth of the water increase?
-
X Corporation was formed and began operations on September 1 of this year. X Corporation expects to have taxable income of $25,000 each quarter of operations for the first two years. X Corporation is...
-
Which of the following exchanges qualify as like-kind exchanges? a. Unimproved land for warehouse b. Factory for apartment building c. Computer for small building d. Inventory for business machine e....
-
Which of the following statements is true regarding the exchange of like-kind property? a. If like-kind property is exchanged and money is paid, a taxable gain or deductible loss must be recognized...
-
Bruce Baxter exchanges an office building worth $1,025,000 for a warehouse owned by Dan Denton. Bruce's adjusted basis in the office building is $750,000 and Dan's basis is $800,000. The fair market...
-
Gerald Golds property is condemned by the local authorities. The property has an adjusted basis of $135,000 and a fair market value of $225,000. The local authorities replace Geralds property with...
-
A particle of mass m , charge q = 1.6 x 10 -19 C and momentum p moves in a circular orbit at a constant speed (in absolute value) in the magnetic field B normal to the orbit. Find the relationship...
-
The nitrogen atoms in N2 participate in multiple bonding, whereas those in hydrazine, N2H4, do not. (a) Draw Lewis structures for both molecules. (b) What is the hybridization of the nitrogen atoms...
-
There are eight different five-carbon alkyl groups. (a) Draw them. (b) Give them systematic names. (c) In each case, label the degree of substitution (primary, secondary, or tertiary) of the head...
-
Use a Newman projection, about the indicated bond, to draw the most stable conformer for each compound. (a) 3-methylpentane about the C2-C3 bond (b) 3, 3-dimethylhexane about the C3-C4 bond
-
(a) Draw the two chair conformations of cis-1, 3-dimethylcyclohexane and label all the positions as axial or equatorial. (b) Label the higher-energy conformation and the lower-energy conformation....
-
why you assume value of Machine A is assumed to decrease by $7,000 each year for the purpose of these calculations?
-
1. You want to create a Dog object. Which of the following would you use to describe it? (Choose all that apply.) A. String breed B. int age C. boolean isADog D. String ownerName E....
-
Part 1: Cathy Forth Photography Services (20 marks) You have been hired by Cathy Forth as her new bookkeeper. She has given you the following information: Use the current date and provide an...

Study smarter with the SolutionInn App


