Identify the hydroxyaldehyde that will cyclize under acidic conditions to give the following hemiacetal: -OH
Question:
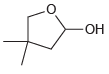
Transcribed Image Text:
-OH
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 77% (9 reviews)

Answered By

Nandana Wijayarathna
I am a highly experienced writer in several areas,
Business management
Information technology
Business administration
Literature
Biology
Environmental science
History
4.50+
161+ Reviews
399+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
When ethylene oxide is treated with a strong nucleophile, the epoxide ring is opened to form an alkoxide ion that can function as a nucleophile to attack another molecule of ethylene oxide. This...
-
The mechanism for acidic hydrolysis of a nitrile resembles the basic hydrolysis, except that the nitrile is first protonated, activating it toward attack by a weak nucleophile (water). Under acidic...
-
Consider the three constitutional isomers of dioxane (C 4 H 8 O 2 ): One of these constitutional isomers is stable under basic conditions as well as mildly acidic conditions and is therefore used as...
-
Below are batting averages of the New York Yankees players who were at bat ve times or more in 2006. (a) Construct a frequency distribution. Explain how you chose the number of bins and the bin...
-
Complete the table below for the missing variances? Total Flexible Budget Product Cost Variance (a) Total Direct Labor Variance (c) Total Manufacturing Overhead Variance Total Direct Materials...
-
Does higher expected inflation increase, decrease, or have no effect on the required rate of return?
-
Consider the multiple regression model fit to the gasoline mileage data in Problem 3.5. Problem 3.5 Consider the gasoline mileage data in Table B.3. a. Construct a normal probability plot of the...
-
BrasPetro S.A., a large Brazilian petrochemical company, reported a balance of R$1,594.9 million in Accounts Receivable at the beginning of Year 7 and R$1,497.0 million at the end of Year 7. Its...
-
3. Write as the sum and/or difference of logarithms. Simplify, if possible. 13 Jab. log (ab)
-
Black Media Inc. owns and operates a large number of news-papers across Canada. On 1 October 20X5, the board of directors voted unanimously to dispose of one of those newspapers, The Daily Con. Black...
-
D-Ribose can adopt two pyranose forms and two furanose forms. (a) Draw both pyranose forms of d-ribose, and identify each as or . (b) Draw both furanose forms of d-ribose, and identify each as or .
-
Draw the cyclic hemiacetal that is formed when each of the following bifunctional compounds is treated with aqueous acid. (a) (b) (c) . H.
-
Explain the difference between the spot (zero-coupon rate) curve and the yield curve.
-
The best way to describe the relationship between the design and manufacturing departments at Waverly Prestressed Concrete (WPC) is warfare . A typical conversation between the vice president for...
-
The FIFA World Cup was one of the biggest sporting events of 2018. The file WC2018Players contains data of the players of the 32 teams that qualified for the event. A dummy variable is included to...
-
You know you have hit the big time when the purple-and orange-lettered Federal Express (FedEx) truck driven by a smiling courier pulls into your driveway or walks up your stairwell and hands you the...
-
Two quality managers meet for lunch every Friday to discuss common problems, compare notes, and make suggestions to each other. Today their discussion has turned into a debate. The topic is the...
-
The Rocky Flats Plant of Rockwell International is committed to hiring hard-core unemployed people for entry-level positions with the intention of migrating them upward. However, many new employees...
-
Crumple zones. A life-saving development in automobile manufacture is the invention of crumple zones, areas of the body and frame of a car deliberately made to collapse in a collision (Fig. P7.64)...
-
1) The government decided to reduce taxes on fast-food to increase revenue. The government assumes that fast-food products have a) An inelastic demand b) An elastic demand c) A demand curve that is...
-
When the following stereoisomer of 2-bromo-1,3-dimethylcyclohexane is treated with sodium methoxide, no E2 reaction is observed. Explain why this compound cannot undergo the E2 reaction in the chair...
-
(a) Two stereoisomers of a bromodecalin are shown. Although the difference between these stereoisomers may seem trivial, one isomer undergoes elimination with KOH much faster than the other. Predict...
-
Give the expected product(s) of E2 elimination for each reaction. (Hint: Use models!) (a) (b) CH, H NaOCH one product HC H NaOCH two products
-
Tim Drinking, Inc. used a predetermined overhead allocation rate to allocate of indirect costs to the Filling Department and the Packaging Department journal entry to record the allocation of...
-
What are the key principles of management, and how do they contribute to effective organizational practices?
-
Discuss the impact of globalization on small businesses in developing countries. Analyze the advantages and disadvantages of globalization for small businesses, and provide recommendations for...

Study smarter with the SolutionInn App


