Identify the number of chirality centers in each of the following compounds: (a) (b) (c) `NH2 N'
Question:
(a)
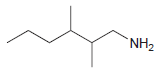
(b)
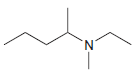
(c)
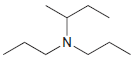
Transcribed Image Text:
`NH2 N'
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 80% (5 reviews)
a T...View the full answer

Answered By

Moses mwangi
With prior writing experience, be sure that I will give a great grade, If not an A+, it will be something close to this. My reviews speaks it all, Try me!!
4.80+
78+ Reviews
157+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
Name the following compounds: a. b. c. d. CH,CH OH Cl Br Br. CH,CH CH3
-
Predict the product(s) for each of the following reactions. In each case, make sure to consider the number of chirality centers being formed. a. b. c. d. e. f. Os0, (catalytic) NMO 1) OsO, 2) NaHSO,...
-
Name the following compounds: a. b. c. d. e. f. Br
-
Determine the degrees of freedom under the following conditions: (a) Tl-20 wt% Pb at 325 C and 400 C; (b) Tl-40 wt% Pb at 325 C and 400 C; (c) Tl-90 wt% Pb at 325 C and 400 C. Refer to the phase...
-
What is the internal rate of return for a project that has a net investment of $75,000 and the following net cash flows: Year 1 = $15,000; Year 2 = $20,000; Year 3 = $25,000; Year 4 = $30,000?
-
Multiple Choice Questions 1. Goods on consignment should be included in the inventory of: a. The consignor but not the consignee b. Both the consignor and the consignee c. The consignee but not the...
-
P(x > 68.4) The random variable x is normally distributed with mean = 74 and standard deviation = 8. Find the indicated probability.
-
F. L. Callans checkbook lists the following: The March bank statement shows Requirement Prepare Callans bank reconciliation at March 31,2015. DateCheck No. Item Check Deposit Balance 640 625 115 740...
-
Suppose the Jenson's alpha of a portfolio is 2.5%, while the actual return is 12%. The risk-free rate is 2.5% and the expected return on the market is 12%. What is the Treynor Ratio of the portfolio?
-
A turbine discharges 200 kg/h of saturated steam at 10.0bar absolute. It is desired to generate steam at 250C and 10.0 bar by mixing the turbine discharge with a second stream of superheated steam of...
-
Do these findings surprise you? Why or why not? How might you apply what you have learned about social media and social media marketing from this chapter to your own workplace ethics?
-
1. Do you agree with P&G that online marketing will save money without hurting its brands in the market place? Why or why not? 2. What non-financial advantages of online marketing can P&G try to...
-
Harris Corporation, a manufacturing of electronics and communications systems, uses a service department charge system to charge profit centers with Computing and Communication Services (CCS) service...
-
DVDs and Blu-ray disks store information in patterns that are read by laser light. The shorter the wavelength of the light, the closer the data tracks can be placed on the disk. A Blu-ray player uses...
-
An investigator is using a laser to illuminate a distant target. He decides that he needs a smaller beam, so he puts a pinhole directly in front of the laser. He finds that this actually spreads the...
-
Describe the meaning and importance of the no-duty-to-treat principle?
-
Explain generally how the U.S. approach to health rights differs from that of other high-income countries?
-
Solar cells generally have an antireflection coating. Explain how this increases their efficiency.
-
Graph each rational function. f(x) x + 1
-
What are some of the possible sources of information about a company that could be used for determining the companys competitive stance?
-
Ammonia appears in Table 1-5 both as an acid and as a conjugate base.
-
Write equations for the following acid-base reactions. Use the information in Table 1-5 to predict whether the equilibrium will favor the reactants or the products. (a) HCOOH + -CH (b) CH3COO- +...
-
Solved Problem 1-5(c) showed protonation of the double-bonded oxygen in acetic acid. Show the product of protonation on the other (-OH) oxygen. Explain why protonation of the double-bonded oxygen is...
-
Victor Korchnoi bought a bond one month before a semi-annual coupon was due. The face value was $10,000 and the coupon rate 8.5%. At the time of purchase there were 34 coupons left and the YTM was 6%...
-
4. Consider a 30-year U.S. Treasury bond paying 4.5 percent coupon, and selling for $1010. What is the yield to maturity? Make sure to show your work. 5. A 30-year U.S. corporate bond with a 6...
-
A project's base case or most likely NPV is $44,000, and assume its probability of occurrence is 50%. Assume the best-case scenario NPV is 65% higher than the base case and assume the worst scenario...

Study smarter with the SolutionInn App


