In each case below, identify the highest and lowest energy conformations. In cases where two or three
Question:
(a)
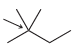
(b)
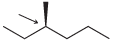
(c)
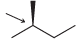
(d)
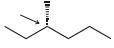
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 75% (12 reviews)
a b c d CH3 CH...View the full answer

Answered By

Branice Buyengo Ajevi
I have been teaching for the last 5 years which has strengthened my interaction with students of different level.
4.30+
1+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
In each case below, draw a Newman projection as viewed from the angle indicated: (a) (b) (c) (d) (e) (f) Observer CI Observer CI
-
In each case below, identify the acid and the base. Then draw the curved arrows showing a proton transfer reaction. Draw the products of that proton transfer, and then predict the position of...
-
In each case below, identify the acid and the base. Then draw the curved arrows showing a proton transfer reaction. Draw the products of that proton transfer, and then predict the position of...
-
Express the given quantity in terms of sin x and cos x. sin 2 X
-
On the basis of the diagram that reflects the effect of imposing a tariff on an importable product, show Effects, calculations and results of the following: 1. Consumption Effect 2 Production Effect...
-
Graph A is the force-versus-time graph for a hockey stick hitting a 160 g puck. Graph Bis the force-versus-time graph for a golf club hitting a 46 g golf ball. Which force delivers the greater...
-
Let $R_{j}^{2}$ be the coefficient of determination when we regress the $j$ th regressor on the other $k-1$ regressors. Show that the $j$ th variance inflation factor may be expressed as...
-
A company issues a $6,000,000, 12%, five-year bond that pays semiannual interest of $360,000 ($6,000,000 12% ), receiving cash of $6,463,304. Journalize the bond issuance.
-
Required Information [The following Information applies to the questions displayed below.] Warnerwoods Company uses a perpetual Inventory system. It entered into the following purchases and sales...
-
For a 70 m long pipeline, 3m and 5m long pipes are used. If the price for shorter pipe is $10 per piece and for the other one $20 per piece, what is the price of the most profitable usage of pipes?
-
For each of the following compounds, predict the energy barrier to rotation (looking down any one of the C-C bonds). Draw a Newman projection and then compare the staggered and eclipsed...
-
Compare the three staggered conformations of ethylene glycol. The anti conformation of ethylene glycol is not the lowest energy conformation. The other two, staggered conformations are actually lower...
-
Stakeholders are less likely to include which of the following groups? A. Owners B. Employees C. Community leaders D. Competitors
-
Calculate real GDP in 2022 expressed in base year prices. Tropical Republic produces only bananas and coconuts. The base year is 2021, and the table gives the quantities produced and the market...
-
Use the following data to work out the year in which the U.S. standard of living (i) increased and (ii) decreased. Explain your answer. Year 2017 2018 2019 2020 Real GDP $18.1 trillion $18.7 trillion...
-
How can information be used to improve peoples decision making? There are several factors in life we cant control and stressing about things like coronavirus and global emergencies isnt unusual. We...
-
Calculate nominal GDP in 2021 and 2022. Tropical Republic produces only bananas and coconuts. The base year is 2021, and the table gives the quantities produced and the market prices. Quantities...
-
What is meant by integrated marketing communications? Explain the advantages of taking an integrated approach to marketing communications.
-
A point on the terminal side of an angle in standard position is given. Find the exact value of each of the six trigonometric functions of . (5, -12)
-
If (x) 0 on the interval [a, b], the definite integral gives the exact area under the curve between x = a and x = b.
-
Explain the following: (a) Cyclononatetraenyl anion is planar (in spite of the angle strain involved) and appears to be aromatic. (b) Although [16]annulene is not aromatic, it adds two electrons...
-
Furan possesses less aromatic character than benzene as measured by their resonance energies (96 kJ mol-1 for furan; 151 kJ mol-1 for benzene). What reaction have we studied earlier that shows that...
-
If benzene were 1,3,5-cyclohexatriene, the carbon-carbon bonds would be alternately long and short as indicated in the following structures. However, to consider the structures here as resonance...
-
1. What is service-oriented architecture and how is it different from Web services architecture? 2. What is the role of the systems approach in the SDLC? 3. Briefly discuss the key phases of the SDLC...
-
1. Define the role of CRM in your own words. 2. What are the key differences between todays CRM and the early generation of CRMs? 3. How does CRM impact the companys bottom line or performance? 4....
-
What do you think are Thorndike's major contributions to learning theory and teaching? Share a time when you have observed Connectionism. How useful do you think the theory of Connectionism is in an...

Study smarter with the SolutionInn App


