In each of the following cases, identify whether the reagent shown is suitable to accomplish the task
Question:
(a) To protonate
using H2O
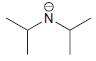
(b) To protonate
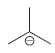
using
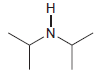
(c) To deprotonate
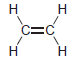
using
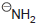
(d) To protonate
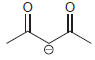
using H2O
(e) To protonate
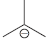
using H2O
(f) To protonate
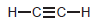
using
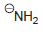
Transcribed Image Text:
OZ
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 66% (12 reviews)
a Yes because a negative charge on an oxygen atom will be more stable than a negative ch...View the full answer

Answered By

Muhammad adeel
I am a professional Process/Mechanical engineer having a vast 7 years experience in process industry as well as in academic studies as a instructor. Also equipped with Nebosh IGC and lead auditor (certified).
Having worked at top notch engineering firms, i possess abilities such as designing process equipment, maintaining data sheets, working on projects, technical biddings, designing PFD and PID's etc.
Having worked as an instructor in different engineering institutes and have been involved in different engineering resrearch projects such as refinery equipment designing, thermodynamics, fluid dynamics, chemistry, rotary equipment etc
I can assure a good job within your budget and time deadline
4.90+
52+ Reviews
60+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
In each of the following cases, calculate the nominal exchange rate assuming purchasing power parity holds. a. A bottle of wine sells for $16 in the United States and 10 in France. b. A book sells...
-
Write balanced net ionic equations for the reactions that occur in each of the following cases. Identify the spectator ion or ions in each reaction. (a) Cr2 (SO4)3(aq) + (NH4)2CO3(aq) (b) Ba...
-
In each of the following cases draw the structure of an alkyl halide that will undergo an E2 elimination to yield only the indicated alkene. a. b. c. d. ? = E2 E2
-
In Exercises show that the function y = (x) is a solution of the differential equation. y = 4e-x y" - y = 0
-
In the Continuing Payroll Problem A, presented at the end of succeeding chapters, you will gain experience in computing wages and salaries and preparing a payroll register for Kipley Company, Inc., a...
-
What do we call the quantity force distance, and what quantity does it change?
-
The study recorded whether participants were female or male \((S e x=F\) or M), and we see that 33 of the 165 females and 42 of the 99 males allowed the pressure to reach its maximum level of \(300...
-
With the rapid growth of cloud computing, many organizations are contracting with third-party service providers to process and store all kinds of data off site. In doing so, entities are now...
-
A company sets an auction for N objects. Bidders place their bids for some subsets of the N objects that they like. The auction house has received n bids, namely bids bj for subset Sj, for j =...
-
According to data from the National Health and Nutrition Examination Survey, 33% of white, 49.6% of black, 43% of Hispanic, and 8.9% of Asian women are obese. In a representative town, 48% of women...
-
As we will learn in Chapter 21, treating a lactone (a cyclic ester) with sodium hydroxide will initially produce an anion: This anion rapidly undergoes an intramolecular proton transfer, in which the...
-
Identify and name the parent in each of the following compounds: (a) (b) (c) (d) (e) (f) (g) (h) (i)
-
The Precision Machining Company makes hand-held tools on an assembly line that produces one product every minute. On one of the products, the critical quality dimension is the diameter (measured in...
-
The following transactions are for Sandhill Company, 1 On December 3, Sandhill Company sold 5647,000 of merchandise to Monty Co., on account, terms 2/10, n/30, FOB destination. Sandhill pald $400 for...
-
Zheng invested $152,000 and Murray invested $252,000 in a partnership. They agreed to share income and loss by allowing a $73,000 per year salary allowance to Zheng and a $53,000 per year salary...
-
How would you implement your public health campaign on teenage vaping . you may explain two potential barriers that might impact the implementation process and one way you might address one barrier....
-
1.What are the main features of the Financial Modernization Act of 1999? 2.What major impact on commercial banking activity occurred from this legislation?
-
Should elders rely on youth to take care of their needs? Does wealth blind people to those that they love? Should both youth and the elderly expect respect from each other?
-
Use the evenodd properties to find the exact value of each expression. Do not use a calculator. sin(-135)
-
Show that, given a maximum flow in a network with m edges, a minimum cut of N can be computed in O(m) time.
-
Propose a reasonable mechanism for the following reaction. OH
-
Propose a reasonable mechanism for the following reaction. OH cat. H PO EtOH
-
Vicinal halo alcohols (halohydrins) can be synthesized by treating epoxides with HX. (a) Show how you would use this method to synthesize 2-chlorocyclopentanol from cyclopentene. (b) Would you expect...
-
What strategies can organizations employ to proactively identify and mitigate budgetary risks, such as cost overruns, revenue shortfalls, and unforeseen contingencies, in order to safeguard financial...
-
Determine if the following piecewise defined function is differentiable at x=0. f(x)= 3x-5, X0 x + 5X-5, X <0 What is the right-hand derivative of the given function? f(0+h)-f(0) lim h h0+ (Type an...
-
Consider the first order differential equation with initial condition t'+3t2y=2t, y(1)=16 where is a function of time t. 1 a. Show that y= + C +3 where C is an arbitrary constant, is the general...

Study smarter with the SolutionInn App


