Predict the major product(s) for each of the following reactions: a. b. c. d. Br2 Br2
Question:
a.
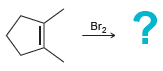
b.
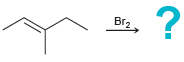
c.
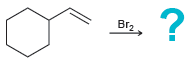
d.
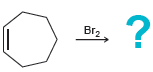
Transcribed Image Text:
Br2 Br2
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 80% (5 reviews)
a b ...View the full answer

Answered By

Shubhradeep Maity
I am an experienced and talented freelance writer passionate about creating high-quality content. I have over five years of experience working in the field and have collaborated with several renowned companies and clients in the SaaS industry.
At Herman LLC, an online collective of writers, I generated 1,000+ views on my content and created journal content for 100+ clients on finance topics. My efforts led to a 60% increase in customer engagement for finance clients through revamping website pages and email interaction.
Previously, at Gerhold, a data management platform using blockchain, I wrote and published over 50 articles on topics such as Business Finance, Scalability, and Financial Security. I managed four writing projects concurrently and increased the average salary per page from $4 to $7 in three months.
In my previous role at Bernier, I created content for 40+ clients within the finance industry, increasing sales by up to 40%.
I am an accomplished writer with a track record of delivering high-quality content on time and within budget. I am dedicated to helping my clients achieve their goals and providing exceptional results.
5.00+
1+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
Predict the major product (or products) formed when each of the following reacts with a mixture of concentrated HNO3 and H2SO4. (a) (b) (c) 4-Chlorobenzoic acid (d) 3-Chlorobenzoic acid (e)...
-
Predict the major product from each of the following reactions. (a) (b) (c) (d) (e) (f) (g) (h) (i) CH NH2, cat. HA N-H, cat. HA NH2 cat. HA PPha (1) HS SH (2) Raney Ni, H2 0 CH2PPha (excess) O. O...
-
Predict the major product from each of the following reactions. (a) (b) (c) (d) (e) HO OH NH2 HA (cat.) (1) HCN (2) LiAIH4 (3) H2o mCPBA
-
.How dependent do you think the success of zero-based budgeting would be on the types of attribution models used? How might you increase the odds of success in this budgeting approach? .Advertising,...
-
What authorizations does the AR Supervisor not have for Account Types? Why do you think the AR Supervisor would be restricted from these authorizations for this accounting document?
-
Consider the potential \(V=-A /\left(x^{2}+a^{2} ight)\), and states \(|1angle,|2angle\), the first two eigenstates in this potential. Using the methods in the text, estimate the transition...
-
Refer to Figure 8, the sequence diagram for the Make Appointment use case. Three spaces are identified in this diagram. Explain the significance of each.
-
The following data pertain to Legion Lighting Companys oak-clad, contemporary chandelier. Variable manufacturing cost.................................................................$300 Applied...
-
Crane Co. has zero units of beginning work in process. During the period, 15,120 units were completed and transferred out, and there were 630 units of ending work in process one-fifth complete as to...
-
In August 2010, Michael Dell, Dell Inc.'s CEO and chairman of the board, was reelected to Dell's board of directors by Dell's shareholders. However, not all of the shareholders were happy with Mr....
-
Compound X has molecular formula C 5 H 10 . In the presence of a metal catalyst, compound X reacts with one equivalent of molecular hydrogen to yield 2-methylbutane. a. Suggest three possible...
-
Predict the major product(s) that are expected when each of the following alkenes is treated with Br 2 /H 2 O: a. b. c. d.
-
The television shows being watched by households surveyed by Nielsen Media Research. Qualitative versus Quantitative Data. In Exercises, determine whether the data described are qualitative or...
-
A payment process that reimburses members for out-of-network health expenses is to be controlled through use of a fraction nonconforming chart. Initially, one sample of size 200 is taken each day for...
-
1. What is the mathematical definition of torque, t? 2. What is the unit of torque, t, in the MKS system of units? 3. Are the units of work and torque the same? 4. What is the basic difference...
-
The large-cap stocks achieved a nominal rate of return of 13.36 percent. The inflationary figure observed over the course of the previous year amounted to 1.61 percent. What is the actual return on...
-
What are the "3 15"and why are these essential to learning in the classroom?
-
Kinross is a major Gold Producer. They are expecting a production of 500,000 oz of Gold in 6 months (they are long Gold). The Forward is : $1,800/ troy oz Kinross Purchases a 6 month Gold Put Struck...
-
Where appropriate, round your answer to the nearest tenth of percent. Determine the answer to the question. Fifty-four is what percent of 600?
-
Read Case Study Google: Dont Be Evil Unless and answer the following: Why do you think Google was adamant about not wanting to supply information requested by the government concerning the Child...
-
Which of the following compounds would you expect to have a ? ? ?* UV absorption in the 200 to 400 nm range? (a) (b) (c) (CH3)2C=C=Do CH2 A ketene Pyridine
-
Predict the products of the following Diels?Alder reactions; (b) (a)
-
Show the structure, including stereochemistry, of the product from the following Diels?Alder reaction: COCH3 C
-
If a tax Bear has a vacation home, that is issues personally by the taxpayer for part of the year, and as a rental for part of the year, how are expenses reported on its tax return?
-
Christopher and Janai have a new grandson. How much money should they invest now so that he will have $78,000 for his college education in 18 years? The money is invested at 3.8% compounded...
-
Jonas' current annual salary is $70,000. Ten years from now how much will he need to earn to retain his present purchasing power if the rate of inflation over that period is 2% /year compounded...

Study smarter with the SolutionInn App


