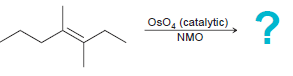
Predict the products of each of the following reactions: a. b. c. d. e. f. g. h.
Question:
a.
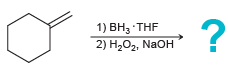
b.
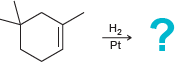
c.
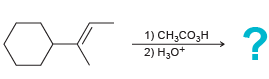
d.
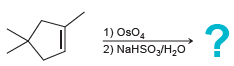
e.
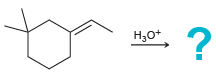
f.
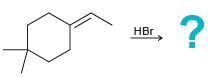
g.
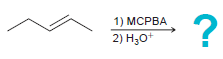
h.
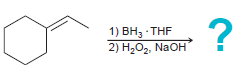
i.
Transcribed Image Text:
1) BH3 THF 2) H2O2, NaOH На Pt
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 69% (13 reviews)
a b c d ...View the full answer

Answered By

JAPHETH KOGEI
Hi there. I'm here to assist you to score the highest marks on your assignments and homework. My areas of specialisation are:
Auditing, Financial Accounting, Macroeconomics, Monetary-economics, Business-administration, Advanced-accounting, Corporate Finance, Professional-accounting-ethics, Corporate governance, Financial-risk-analysis, Financial-budgeting, Corporate-social-responsibility, Statistics, Business management, logic, Critical thinking,
So, I look forward to helping you solve your academic problem.
I enjoy teaching and tutoring university and high school students. During my free time, I also read books on motivation, leadership, comedy, emotional intelligence, critical thinking, nature, human nature, innovation, persuasion, performance, negotiations, goals, power, time management, wealth, debates, sales, and finance. Additionally, I am a panellist on an FM radio program on Sunday mornings where we discuss current affairs.
I travel three times a year either to the USA, Europe and around Africa.
As a university student in the USA, I enjoyed interacting with people from different cultures and ethnic groups. Together with friends, we travelled widely in the USA and in Europe (UK, France, Denmark, Germany, Turkey, etc).
So, I look forward to tutoring you. I believe that it will be exciting to meet them.
3.00+
2+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
Give the products of each of the following reactions: a. b. c. d. e. f. g. h. i. j. HCI CH2CH3 1. CH3CH2MgBr 1. CH3CH2MgBr excesS CH,CH,COCH 2. H20 ot.cum 1. LiAIH4 NO 2. H20 catalytic Ht + CH...
-
Give the product of each of the following reactions: a. b. c. d. e. f. g. h. CH CH3 CH CH H2S04 CH,CH CHCH CH ,504 CH CH-CCH CH3 OH ' CH3O CH3OH 1. TsCl/pyridine 2. NaCEN CH3 OH (CH2CH2CH2hCuli CI
-
Give the product of each of the following reactions: a. b. c. d. e. f. g. h. hv CH3 H3C CH3 A DHHD H3C
-
Using algebra, find the slope and y-intercept of the line represented by each of the following equations. (a) 7x + 3y = 6 (b) 10y = 5x (c) (2y 3x)/2 = 4 (d) 1.8x + 0.3y 3 = 0 (e) 1/3x = -2 (f) 11x...
-
The following activities were observed for a mold operator: a) operator removes molded piece from die, b) walks to bench, puts the piece into box and the puts the box on a pallet, c) returns to...
-
You are trying to estimate the beta of a private firm that manufactures home appliances. You have managed to obtain betas for publicly traded firms that also manufacture home appliances. The private...
-
Comparative data extracted from the general purpose financial statements and notes thereto of Express Delivery Ltd are presented below. Required (a) Prepare a trend analysis of the data. (b) Comment...
-
Braxton Technologies, Inc., constructed a conveyor for A&G Warehousers that was completed and ready for use on January 1, 2011. A&G paid for the conveyor by issuing a $100,000, four-year note that...
-
ASE STUDY APPLICANT TESTING AT THE RCMP Applicants to the Royal Canadian Mounted Police (RCMP) must pass a written examination, an inter- view, and a physical ability test before being accepted for...
-
Based on Problem 2-9B, prepare a statement of owners equity for David Segal for the month of October 20--.
-
Why is a correlation c oefficient never greater than 1 (or less than 1)?
-
The rate at which two methyl radicals couple to form ethane is significantly faster than the rate at which two tert-butyl radicals couple. Offer two explanations for this observation.
-
1. In what sense does goal setting play a role in Barcelona's methods for enhancing performance? Judging from the video, how would you respond to the following questions: What kind of goals does...
-
OJ Ltd has been having difficulty collecting its accounts receivable. For the year 2016, the company increased the allowance for doubtful accounts by $48 000, bringing the balance to $70 000. At the...
-
In each of the following cases, what component of good internal control is being violated (if any)? 1. Tough Ltd pays all its employees minimum wages and does not have pleasant working conditions. 2....
-
Briefly explain what is meant by the statement: 'Under historic cost accounting, depreciation is a process of allocation.' Include in your answer some reference to the key assumptions that support...
-
XYZ commenced operations on 1 June 2016 selling one type of shirt. The company uses FIFO (first in, first out) and perpetual inventory control. The June inventory and sales records for the shirts...
-
Eaglehawk Ltd had the following noncurrent asset on its balance sheet on 30 June 2015. The company adopts a policy of depreciating all relevant items on a straight-line basis at an annual rate of 10...
-
Determine the following indefinite integrals. Check your work by differentiation. ? r2 dx * 4x4
-
The words without recourse on an indorsement means the indorser is: a. not liable for any problems associated with the instrument. b. not liable if the instrument is dishonored. c. liable personally...
-
Draw compounds that meet these requirements: (a) A primary alcohol (b) A tertiary alcohol (c) A secondary alkyl chloride (d) A secondary amine
-
What is wrong with the name given for these compounds? Provide the correct name for each. a) c) 18 2-Ethyl-2-pentene CHCHCH 3-Cyclohexylpropane 1-Chloro-2-cyclopentene OH 2-2-Methylbutylcyclopentanol...
-
Name these compounds: a) ) g) 1) k) - OH NH b) CI d) f) CH=CHCHC=CH h) Br j) O OCH 3
-
What is the balance in Work in Process Inventory at the end of each month? Work in Process Inventory April 30 $ May 31 A 10,140 15,950 June 30 $ 10,450
-
Sako Company s Audio Division produces a speaker used by manufacturers of various audio products. Sales and cost data on the speaker follow: Selling price per unit on the intermediate market $ 6 0...
-
Vulcan Company's contribution format income statement for June is as follows: Vulcan Company Income Statement For the Month Ended June 30 Sales Variable expenses Contribution margin Fixed expenses...

Study smarter with the SolutionInn App


