Predict the value for the specific rotation of the following compound. Explain your answer. - , .
Question:
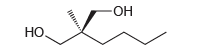
Transcribed Image Text:
-ОН но, Но.
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 63% (11 reviews)
This compound does not have a ...View the full answer

Answered By

PALASH JHANWAR
I am a Chartered Accountant with AIR 45 in CA - IPCC. I am a Merit Holder ( B.Com ). The following is my educational details.
PLEASE ACCESS MY RESUME FROM THE FOLLOWING LINK: https://drive.google.com/file/d/1hYR1uch-ff6MRC_cDB07K6VqY9kQ3SFL/view?usp=sharing
3.80+
3+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
The specific rotation of the R enantiomer of the following alkene is a20D = +76 degree mL g-1 dm-1, and its molecular mass is 146.2. CH HC CHC CH Ph
-
The observed rotation of 2.0 g of a compound in 50 mL of solution in a polarimeter tube 50-cm long is +13.4o What is the specific rotation of the compound?
-
The compound pictured in the margin is a sugar called (-)arabinose. Its specific rotation is -105. (a) Draw the enanfiomer of (-)-arabinose. (b) Does (-)-arabinose have any other enantiomers? (c)...
-
Find the lengths of the curves. The cardioid r = 1 + cos
-
Contrast the work of Renoir and Laurtrec. How do the subjects' styles of the artists reflect nineteenth century French society and the innovations of nineteenth century art? Use examples to support...
-
The table shows the heights (in inches) and weights (in pounds) of 14 college men. The scatterplot shows that the association is linear enough to proceed. a. Find the equation for the regression line...
-
Compare the total surface area of a group of N small-diameter spherical particles with that of a single large-diameter spherical particle having the same volume.
-
Suppose Lattin Corp.s breakeven point is revenues of $1,500,000. Fixed costs are $720,000. Required 1. Compute the contribution margin percentage. 2. Compute the selling price if variable costs are $...
-
Wardell Company purchased a minicomputer on January 1, 2022, at a cost of $57,000. The computer was depreciated using the straight-line method over an estimated five-year life with an estimated...
-
A company sells precision grinding machines to four customers in four different countries. It has just signed a contract to sell, two months from now, a batch of these machines to each customer. The...
-
When 1.30 g of menthol is dissolved in 5.00 mL of ether and placed in a sample cell 10.0 cm in length, the observed rotation at 20C (using the D line of sodium) is +0.57. Calculate the specific...
-
Identify whether each of the following compounds exhibits a molecular dipole moment. For compounds that do, indicate the direction of the net molecular dipole moment: a. CHCl 3 b. CH 3 OCH 3 c. NH 3...
-
Suppose that the mean download time for a commercial tax preparation site is 2.0 seconds. Suppose that the download time is normally distributed, with a standard deviation of 0.5 second. What is the...
-
A profit maximum is least likely to occur when: A. average total cost is minimized. B. marginal revenue equals marginal cost. C. the difference between total revenue and total cost is maximized.
-
The slope of a supply curve is most often: A. zero. B. positive. C. negative.
-
What role does the opportunity cost rate play in calculating financial returns?
-
Consider these two quotes concerning recent Federal Reserve policy On December 12, 2012 the Federal Reserve issued the following statement: "In particular, the Committee decided to keep the target...
-
Is the calculation of financial return an application of time value analysis? Explain your answer.
-
The United States imports about half of its petroleum needs. Suppose that the rest of the oil producers are willing to supply as much oil as the United States wants at a constant price of $25 a...
-
Halley's comet travels in an ellipti- cal orbit with a = 17.95 and b = 4.44 and passes by Earth roughly every 76 years. Note that each unit represents one astronomical unit, or 93 million miles. The...
-
Base-promoted hydrolysis of methyl mesitoate occurs through an attack on the alcohol carbon instead of the acyl carbon: (a) Can you suggest a reason that accounts for this unusual behavior? (b)...
-
What products would you obtain from acidic and basic hydrolysis of each of the following amides? (a) N, N-Diethylbenzamide (b) (c) NH2 HO (a dipeptide) CeHs
-
(a) Provide the reagents required to accomplish the following transformation. (b) What product would you likely obtain if you attempted to synthesize the nitrile above by the following method? CO,H...
-
An 90000 loan is amortized by payments of $1850 at the end of every 6 months at a rate of 2% compounded monthly 1. Construct a partial amortization schedule showing the last 2 payments 2. determine...
-
Kelso's has a return on equity of 15.2 percent, a debt-equity ratio of 44 percent, a capital intensity ratio of 1.08, a current ratio of 1.25, and current assets of $138,000. What is the profit...
-
The following condensed information was reported by Peabody Toys, Incorporated, for 2024 and 2023: Income statement information Net sales Net income Balance sheet information Current assets Property,...

Study smarter with the SolutionInn App


