The specific rotation of the R enantiomer of the following alkene is a20D = +76 degree mL
Question:
The specific rotation of the R enantiomer of the following alkene is a20D = +76 degree mL g-1 dm-1, and its molecular mass is 146.2.
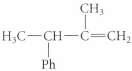
Transcribed Image Text:
CH HC CHC CH Ph
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 53% (13 reviews)
The rotation of the enantiomer cancels h...View the full answer

Answered By

JAPHETH KOGEI
Hi there. I'm here to assist you to score the highest marks on your assignments and homework. My areas of specialisation are:
Auditing, Financial Accounting, Macroeconomics, Monetary-economics, Business-administration, Advanced-accounting, Corporate Finance, Professional-accounting-ethics, Corporate governance, Financial-risk-analysis, Financial-budgeting, Corporate-social-responsibility, Statistics, Business management, logic, Critical thinking,
So, I look forward to helping you solve your academic problem.
I enjoy teaching and tutoring university and high school students. During my free time, I also read books on motivation, leadership, comedy, emotional intelligence, critical thinking, nature, human nature, innovation, persuasion, performance, negotiations, goals, power, time management, wealth, debates, sales, and finance. Additionally, I am a panellist on an FM radio program on Sunday mornings where we discuss current affairs.
I travel three times a year either to the USA, Europe and around Africa.
As a university student in the USA, I enjoyed interacting with people from different cultures and ethnic groups. Together with friends, we travelled widely in the USA and in Europe (UK, France, Denmark, Germany, Turkey, etc).
So, I look forward to tutoring you. I believe that it will be exciting to meet them.
3.00+
2+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Organic Chemistry questions
-
(a) Draw line-and-wedge structures for the two enantiomers of the following allene. (b) One enantiomer of this compound has a specific rotation of - 30.7o. What is the specific rotation of the other?...
-
What observed rotation is expected when a 1.5 M solution of (R)-2-butanol is mixed with an equal volume of a 0.75 M solution of racemic 2-butanol, and the resulting solution is analyzed in a sample...
-
(a) The specific rotation of sucrose (table sugar) in water is 66.5 degrees mL g-1 dm-1. What is the observed optical rotation in a 1 dm path of a sucrose solution prepared from 5 g of sucrose and...
-
On January 1, 2019, Chiz Company acquired equipment to be used in its manufacturing operations. The equipment has an estimated useful life of 10 years and an estimated residual value of P50,000. The...
-
What are the probable effects on sales and profits of each of the following credit these policies?
-
Journalize and post the entries required by the following transactions for Francisco Furniture Repair Company. Prepare a trial balance as of April 30, 20X0, for the period April 1 to April 30, 20X0....
-
Suppose \(V=V(S)\). Find the most general solution of the Black-Scholes equation.
-
The December 31, 2020, unadjusted trial balance for Musical Sensations after its second year of operations follows: Required 1. Enter the unadjusted trial balance onto a work sheet. 2. Using the...
-
Mason Company provided the following data for this year: Sales Direct labor cost Raw material purchases Selling expenses Administrative expenses Manufacturing overhead applied to work in process...
-
As the auditor of Clearwater County you learn that various assets are subject to spending constraints. Indicate how each of the following constraints would affect the countys reported fund balance...
-
(a) Draw sawhorse projections of ephedrine (Problem 6.32) about the C1-C2 bond for all three staggered and all three eclipsed conformations. (b) Examine each conformation for chirality. How do the...
-
What is the absolute configuration of (+) -methyl- hexane if catalytic hydrogenation of (S)-(+)-3- methyl-1-hexene gives (-)-3-methylhexane?
-
The LP model of Problem 4(d), Set 4.4a, has no bounded solution. Show how this condition is detected by the generalized simplex procedure.
-
Kaplan & Norton suggest techniques for implementing strategies without disrupting organizations. Provide examples from your work experience of disruptive and non-disruptive strategy implementations. ...
-
explain how to respond to the post below in an educated manner? Hello everyone, I hope everyone is having a lovely week. The positive thing that happened to me this week is that my car play is...
-
The UDL and the COE lesson plan align by ensuring that each student is thought of and multiple engagement strategies of teaching are involved in each lesson. Section 1:Multiple Means of Engagement I...
-
Part A: Safety and Security in the Workplace Discuss some of the common safety and security concerns in today's workplace. What are some important laws that may help organizations reduce risk? What...
-
One characteristic of inclusive leadership is respect for diverse cultural preferences and beliefs. To what extent should this govern the style and behavior of the inclusive leader? Why?
-
Amber is a shareholder of Southern Ferns Ltd. The companys return on ordinary shareholders equity increased in 2016, but Amber received less dividends. Explain to Amber how this can occur.
-
A regular deposit of $100 is made at the beginning of each year for 20 years. Simple interest is calculated at i% per year for the 20 years. At the end of the 20-year period, the total interest in...
-
Draw a conventional structure corresponding to the following skeletal structure, and then name it.
-
Which of the following alkenes can exist as double-bond stereoisomers? Identify the stereocenters in each. (a) H,C=CHCH,CH,CH, (b) CH,CH,CH=CHCH,CH, 1-pentene 3-hexene (c) HC=CH-CH=CH-CH3 (d)...
-
Name the following compound using IUPAC substitutive nomenclature. HC=CCH,CH,CH, I CH,CH,CH,CH,CH,
-
a) Based on the information provided in the table below: i. Describe the impact of the cost behavior on economies of scale, constant returns to scale, and diseconomies of scale in each firm?...
-
Use the following information to answer questions below: Total current assets Total non-current assets Total current liabilities Long-term borrowings Share capital Revenue Gross profit Retained...
-
A Public Information Officer ( PIO ) for a public safety or emergency management agency might expect to spend what portion of his or her time on activities directly related to incident response or...

Study smarter with the SolutionInn App


