The following are mass spectra for the constitutional isomers ethylcyclohexane and 1, 1-dimethylcyclohexane. Based on likely fragmentation
Question:
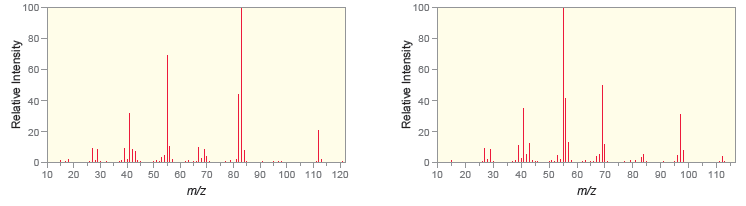
Transcribed Image Text:
100 100 80 - 80- 60 - 60- 40- 40- 20- 0+ 10 100 110 120 90 40 50 60 70 80 20 30 40 50 60 70 80 100 110 10 30 90 m/z m/z
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 78% (14 reviews)
In the first spectrum the base peak appears at M29 signifying ...View the full answer

Answered By

Jacob Festus
I am a professional Statistician and Project Research writer. I am looking forward to getting mostly statistical work including data management that is analysis, data entry using all the statistical software’s such as R Gui, R Studio, SPSS, STATA, and excel. I also have excellent knowledge of research and essay writing. I have previously worked in other Freelancing sites such as Uvocorp, Essay shark, Bluecorp and finally, decided to join the solution inn team to continue with my explicit work of helping dear clients and students achieve their Academic dreams. I deliver, quality and exceptional projects on time and capable of working under high pressure.
4.90+
1252+ Reviews
2844+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
Below are mass spectra for four different compounds. Identify whether each of these compounds contains a bromine atom, a chlorine atom, or neither. a. b. c. d. 100- 80- 60- 60- 40 20- 0- 60 70 10 20...
-
The IR and mass spectra for three different compounds are shown in Figures 13.45-13.47. Identify each compound. a. b. c. 25 26 2.7 28 19 2000 100 43 58 100 71 85 0 20 40 60 80 100 120 m/z 13 14 15 6...
-
For each molecular formula, draw all the possible constitutional isomers of alcohols with that formula. Give the IUPAC name for each alcohol. (a) C3H8O (b) C4H10O (c) C3H6O (d) C3H4O
-
In a given week, 12 babies are born in hospital. Assume that this sample came from an underlying normal population. The length of each baby is routinely measured and is listed below (in cm): 49, 50,...
-
On July 1, 2013, Pizza Company decided to trade-in their used equipment (ovens, refrigerators, ect.) for new models at Sears. The old equipment was initially purchased for $120,000 in January 2010....
-
Use the Lippmann-Schwinger equation for \(\psi\) to write an expression for \(\sigma_{l}\) in terms of integrals involving the potential \(V(r)\) and the wave function.
-
For each of the following situations, calculate the standard error of the mean \(s \mathrm{X}^{-}\). a. \(s=5.00 ; N=16\) b. \(s=17.82 ; N=10\) c. \(s=2.31 ; N=37\) d. \(s=51.32 ; N=21\)
-
The financial statements for Tootsie Roll Industries appear in Appendix A at the end of this book. Instructions Answer these questions using the Consolidated Income Statement . (a) What was the...
-
A manufacturing company reports the following for the period: Inventories Beginning Ending Raw materials $ 1 8 , 1 2 0 $ 1 2 , 1 0 0 Work in process 9 , 5 0 0 1 1 , 3 0 0 Finished goods 1 2 , 5 6 0...
-
Which of the isomeric C4H10O alcohols can be prepared by hydrogenation of aldehydes? Which can be prepared by hydrogenation of ketones? Which cannot be prepared by hydrogenation of a carbonyl...
-
Identify two peaks that are expected to appear in the mass spectrum of 3-pentanol. For each peak, identify the fragment associated with the peak, and show a mechanism for its formation.
-
How would you distinguish between each pair of compounds using high-resolution mass spectrometry? a. b. .
-
Christi, Inc., is using a costs-of-quality approach to evaluate design engineering efforts for a new skateboard. Christis senior managers expect the engineering work to reduce appraisal, internal...
-
Joe owns a bond which is callable in 3 years. The bond has a 6 percent coupon, pays interest semiannually, has a par value of $1,000, and has a yield to call of 6.3 percent. What is the call premium...
-
Compare and contrast the somatic and autonomic division of the peripheral nervous system.
-
If your proposed grade seems too high or too low, your instructor will review the data available (attendance logs, quiz completion, your assignments, etc.) and may adjust your proposed grade up or...
-
Instructions Skandinaviska Enskilda Banken (SEB), one of northern Europe's largest banking groups, operates in several countries, including Germany, Poland, and Russia. In the past, many of SEB's...
-
Northern Metal Manufacturing (NMM), your company, has four manufacturing plants. Sales have been poor lately due to the weak economy that has resulted from the COVID-19 pandemic. Your manager, Marcia...
-
The table in the left column at the top of the next page shows the five most frequently visited news websites in October, 2014. Website.................................Number of Visitors Yahoo! News...
-
An investor sells a European call on a share for $4. The stock price is $47 and the strike price is $50. Under what circumstances does the investor make a profit? Under what circumstances will the...
-
Predict the structure of the product of this reaction: The product has no infrared absorption in the 1620-1680-cm-1 region. NaOH in aqueous EtOH CI HS
-
Cis-4-Bromocyclohexanol racemic C6H10O (compound C) Compound C has infrared absorption in the 1620-1680-cm-1 and in the 3590-3650-cm-1 regions. Draw and label the (R) and (S) enantiomers of product C.
-
1-Bromo [2.2.1] bicycloheptane is unreactive toward both SN2 and SN1 reactions. Open the computer molecular model at the book's website titled "1-Bromo [2.2.1] bicycloheptane" and examine the...
-
What is one way in which a leader can apply the "Status" part of the SCARF model to trigger the "Reward" response?
-
Complete the table to assess risks and develop contingencies for resources you are planning for. Potential risks Planned Response Risk associated with the acquisition or implementation of the...
-
Assignment: persuasive essay for or against Andrew Jackson. In you essay answer the question "Was he a "people's president" or was he more like a king?" Documentary:...

Study smarter with the SolutionInn App


