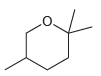
The following cyclic ether can be prepared via an intramolecular Williamson ether synthesis. Show what reagents you
Question:
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 64% (14 reviews)
Ho...View the full answer

Answered By

Joseph Mwaura
I have been teaching college students in various subjects for 9 years now. Besides, I have been tutoring online with several tutoring companies from 2010 to date. The 9 years of experience as a tutor has enabled me to develop multiple tutoring skills and see thousands of students excel in their education and in life after school which gives me much pleasure. I have assisted students in essay writing and in doing academic research and this has helped me be well versed with the various writing styles such as APA, MLA, Chicago/ Turabian, Harvard. I am always ready to handle work at any hour and in any way as students specify. In my tutoring journey, excellence has always been my guiding standard.
4.00+
1+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
Each of the following alcohols can be prepared via reduction of a ketone or aldehyde. In each case, identify the aldehyde or ketone that would be required. a. b. c. d. .
-
Each of the following alcohols can be prepared via reduction of a ketone or aldehyde. In each case, identify the aldehyde or ketone that would be required. a. b. c. d. .
-
Each of the following alcohols can be prepared via reduction of a ketone or aldehyde. In each case, identify the aldehyde or ketone that would be required. a. b. c. d. HO. HO.
-
Which of the following is not necessary to do before you can run a Java program? a. Coding b. Compiling c. Debugging d. Saving
-
Salmon Inc. has debt with both a face and a market value of $3,000. This debt has a coupon rate of 7% and pays interest annually. The expected earnings before interest and taxes is $1,200, the tax...
-
Two social workers just out of college join the same social welfare agency. Both find their caseloads very heavy and their roles very ambiguous. One exhibits negative stress reactions, including...
-
Consider an underlying linear model, \(y_{i}^{*}=\mathbf{x}_{i}^{\prime} \boldsymbol{\beta}+\epsilon_{i}^{*}\), where \(\epsilon_{i}^{*}\) is normally distributed with mean zero and variance...
-
It was announced today that Florida, Inc., will acquire Menlo Park, Inc. Menlo Park has assets with a gross tax basis of $ 6 million and has $ 1.5 million of liabilities. Prior to being acquired,...
-
14 Complete this question by entering your answers in the tabs below. 00:30:48 Required 1 Required 2 Prepare journal entries to record the following transactions for Sherman Systems. a. Purchased...
-
Each year forbes ranks the world's most valuable brands. A portion of the data for 82 of the brands in the 2013 forbes list is shown in Table 2.12 (forbes website, February, 2014). The data set...
-
The heat capacity of α -quartz is given by The coefficient of thermal expansion is given by β = 0.3530 à 10 4 K 1 and V m = 22.6 cm 3 mol+. Calculate ÎS m for...
-
Does the enthalpy of formation of H 2 O(l) change if the absolute enthalpies of H 2 (g) and O 2 (g) are set equal to 100. kJ mol 1 rather than to zero? Answer the same question for CO 2 (g). Will H o...
-
Assuming the yield to maturity remains constant, what is the price of the bond immediately before it makes its first coupon payment?
-
3. Consider the monthly returns of McDonald's stock from August 1966 to December 2014. The data are available from CRSP and in the file m-mcd3dx6614.txt. Obtain the log return series of MCD stock....
-
Gustav bought a zero coupon bond in 2010 for $485.19. In 2020, he redeemed it for $1,000. What was his internal rate of return on this investment as a percentage to one decimal place?
-
How would one monitor if the stocks in their portfolio contain SRI companies or would you just rely on the investment manager (with fingers crossed)? Do you think the average investor is savvy enough...
-
Alliant (a utility company that purchases natural gas) is wanting to hedge its exposure to fluctuations in the price of Natural gas between now and March 2023. Natural gas is sold in units called...
-
On January 1, 2025, Vaughn Company purchased a new machine for $4190000. The new machine has an estimated useful life of nine years, and the salvage value was estimated to be $140000. Depreciation...
-
The expenses for Jorge, a real estate agent, to list, advertise, and attempt to sell a house are $1400. If Jorge succeeds in selling the house, he will receive a commission of 6% of the sales price....
-
Extend Algorithms 3.4 and 3.5 to include as output the first and second derivatives of the spline at the nodes.
-
Amine bases in nucleic acids can react with alkylating agents in typical SN2 reactions. Look at the following electrostatic potential maps, and tell which is the better nucleophile, guanine or...
-
Human brain natriuretic peptide (BNP) is a small peptide of 32 amino acids used in the treatment of congestive heat failure. How many nitrogen bases are present in the DNA that codes for BNP?
-
Human and horse insulin both have two polypeptide chains, with one chain containing 21 amino acids and the other containing 30 amino acids. They differ in primary structure at two places. At position...
-
Details of the capital structure of Webber Ltd. appear below: Bonds Number issued Coupon rate Interest payments Years to maturity 27,000 6% semi-annually 10 $90 Current price Preferred shares Number...
-
An Ice cream company has commissioned an ice sculpture for their end of year celebration. The ice sculpture is in the shape of a giant ice cream cone-the shape can be thought of as a cone with one...
-
1. A soft drink vendor at a popular beach resort analyzes his sales records and finds that if he seels x cans of pop in one day, his profit in dollars is given by the function P(x) = -0.012+3x-80....

Study smarter with the SolutionInn App


