The heat capacity of α -quartz is given by The coefficient of thermal expansion is given by
Question:
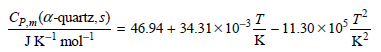
The coefficient of thermal expansion is given by β = 0.3530 × 10ˆ’4 Kˆ’1 and Vm = 22.6 cm3 mol+. Calculate ΔSm for the transformation α -quartz (15.0°C,1atm) †’α -quartz (420.°C, 925 atm).
Transcribed Image Text:
Cp m (a-quartz, s) Jк'mol- 46.94 + 34.31x10-3 - 11.30 x10°, к? к
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 71% (7 reviews)
ASm I Cpm VBPs P dT T 69315 28815 46943431101 113x105 10 1 ...View the full answer

Answered By

Saikumar Ramagiri
Financial accounting:- Journal and ledgers, preparation of trail balance and adjusted trail balance Preparation of income statement, retained earning statement and balance sheet Banks reconciliation statements Financial statement analysis Cash flow statement analysis (both direct and indirect methods) All methods of Depreciations Management Accounting:- Ratios Budgeting control Cash budget and production budget Working capital management Receivable management Costing:- Standard and variance costing Marginal costing and decision making Cost-volume-profit analysis Inventory management (LIFO, FIFO) Preparation and estimation of cost sheet Portfolio management:- Calculation of portfolio standard deviation or risk Calculation of portfolio expected returns CAPM, Beta Financial management:- Time value of money Capital budgeting Cost of capital Leverage analysis and capital structure policies Dividend policy Bond value calculations like YTM, current yield etc International finance:- Derivatives Futures and options Swaps and forwards Business problems Finance problems Education (mention all your degrees, year awarded, Institute/University, field(s) of major): Education Qualification Board/Institution/ University Month/Year of Passing % Secured OPTIONALS/ Major ICWAI(inter) ICWAI inter Pursuing Pursuing - M.com(Finance) Osmania University June 2007 65 Finance & Taxation M B A (Finance) Osmania University Dec 2004 66 Finance & Marketing. B.Com Osmania University June 2002 72 Income Tax, Cost & Mgt, Accountancy, Auditing. Intermediate (XII) Board of Intermediate May 1999 58 Mathematics, Accountancy, Economics. S S C (X) S S C Board. May 1997 74 Mathematics, Social Studies, Science. Tutoring experience: • 10 year experience in online trouble shooting problems related to finance/accountancy. • Since 6 Years working with solution inn as a tutor, I have solved thousands of questions, quick and accuracy Skills (optional): Technical Exposure: MS Office, SQL, Tally, Wings, Focus, Programming with C Financial : Portfolio/Financial Management, Ratio Analysis, Capital Budgeting Stock Valuation & Dividend Policy, Bond Valuations Individual Skills : Proactive Nature, Self Motivative, Clear thought process, Quick problem solving skills, flexible to complex situations. Achievements : 1. I have received an Award certificate from Local Area MLA for the cause of getting 100% marks in Accountancy during my Graduation. 2. I have received a GOLD MEDAL/Scholarship from Home Minister in my MBA for being the “Top Rank student “ of management institute. 3. I received numerous complements and extra pay from various students for trouble shooting their online problems. Other interests/Hobbies (optional): ? Web Surfing ? Sports ? Watching Comics, News channels ? Miniature Collection ? Exploring hidden facts ? Solving riddles and puzzles
4.80+
391+ Reviews
552+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
The heat capacity of anhydrous potassium hexacyanoferrate (II) varies with temperature as follows: TIK Cp,m/J K-I mol-I) TIK Cp,m/JK-I mol-I) 10 2.09 100 179.6 20 14.43 110 192.8 30 36.44 150 237.6...
-
Two formulas for the heat capacity of CO are given here: Cp [cal/(mol C)] = 6.890 + 0.001436T (C) Cp (Btu/(lb-moleF)] = 6.864 + 0.0007978T (F) Starting with the first formula, derive the second....
-
A small pharmaceutical firm plans to manufacture a new drug and has hired you as a consultant to design a condenser to remove the drug from a gasvapor mixture. The mixture, which contains 20 mole% of...
-
Wical Rental Management Services manages four apartment buildings, each with a different owner. Wicals CEO has observed that the apartment buildings with more expensive rental rates tend to require...
-
Spice Inc.'s unit selling price is $60, the unit variable costs are $35, fixed costs are $125,000 and current sales are 10,000 units. How much will operating income change is sales increase by 8,000...
-
Imagine that a person who greatly dislikes bureaucracy assumes her first job as an investigator in a very bureaucratic government tax office. Describe the stressors that she might encounter in this...
-
Under the random utility interpretation, an individual with utility \(U_{i j}=\) \(u_{i}\left(V_{i j}+\epsilon_{i j} ight)\), where \(j\) may be 1 or 2 , selects category corresponding to \(j=1\)...
-
You are selling a product on commission, at the rate of $1,000 per sale. To date you have spent $800 promoting a particular prospective sale. You are confident you can complete this sale with an...
-
Current Attempt in Progress Pharoah Industries sells two electrical components with the following characteristics. Fixed costs for the company are $390,000 per year. XL-709 CD-918 Sales price $28 $43...
-
Which of the graphs in Fig. Q25.12 best illustrates the current I in a real resistor as a function of the potential difference V across it? Explain. Figure Q25.12 (a) (b) (c) (d)
-
Calculate the average CH bond enthalpy in methane using the data tables. Calculate the percent error in equating the average CH bond energy in Table 4.3 with the bond enthalpy. Table 4.3 Selected...
-
The following cyclic ether can be prepared via an intramolecular Williamson ether synthesis. Show what reagents you would use to make this ether.
-
A unity feedback system has the loop transfer function (a) Determine the phase margin of the system when K = 0.16. (b) Use the phase margin to estimate £ and predict the overshoot, (c)...
-
Calla Canoe Company has experienced rapid growth in its first few months of operations and has had a sig in customers renting canoes and purchasing T-shirts. Many of these customers are asking for...
-
Lost A Friend, Inc. sells its product at a price of $24 per unit and has $15 in per unit variable costs. The company's total fixed costs are $183,500. What is the firm's break-even point in units...
-
Wall Company bought a trademark from Black Corporation on January 1 for $112,000. An independent consultant retained by Wall estimated that the remaining useful life is 50 years. Its unamortized cost...
-
Use the table to explain methods of preventing and treating each of these STIS/STDs. (6 points) Human papilloma virus Chlamydia Trichomoniasis Gonorrhea Syphilis HIV/AIDS Prevention Treatment...
-
Describe the primary mission of the social work profession. For your response, imagine you are describing this to a friend or family member. Define what constitutes an ethical dilemma. Give an...
-
Assume that you are blind-folded and throw a dart at the dart board shown below. Assume that your dart sticks in the dart board, and not on a line. (a) Determine the probabilities that the dart lands...
-
Let (X. A. p) be a measure space. Show that for any A,B A, we have the equality: (AUB)+(An B) = (A) + (B).
-
In which of the following systems is the energy level separation the largest? (a) A 14Nnucleus in (for protons) a 600 MHz NMR spectrometer, (b) An electron in a radical in a field of 0.300 T
-
Calculate the magnetic field needed to satisfy the resonance condition for unshielded protons in a 150.0 MHz radiofrequency field.
-
Use Table 15.2 to predict the magnetic fields at which (a) 14N, (b) 19F, and (c) 31p comes into resonance at (i) 300 MHz, (ii) 750 MHz.
-
A retirement home in Florida costs $ 2 0 0 , 0 0 0 today. Housing prices in Florida are increasing at a rate of 4 % per year. Joe wants to buy the home in 8 years when he retires. Joe has $ 2 5 , 0 0...
-
1. (55 points) Answer the following questions about the steady state system shown below. (Don't worry about starting from 1st Law - use common engineering assumptions.) a) Is it appropriate to use...
-
Describe at least three ways that Guidance Software responds to and helps to manage changes in our technology - driven world? Describe what forecasting involves and list three limitations to...

Study smarter with the SolutionInn App


