Using ethanol as your only source of carbon atoms, propose a synthesis for each of the following
Question:
(a)
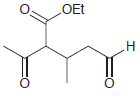
(b)
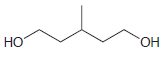
Transcribed Image Text:
OEt .Н но НО он
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 53% (15 reviews)
a b OH PCC CHCl H NaC...View the full answer

Answered By

Ayush Mishra
I am a certified online tutor, with more than 3 years of experience in online tutoring. My tutoring subjects include: Physics, Mathematics and Mechanical engineering. I have also been awarded as best tutor for year 2019 in my previous organisation. Being a Mechanical Engineer, I love to tell the application of the concepts of science and mathematics in the real world. This help students to develop interest and makes learning fun and easy. This in turn, automatically improves their grades in the subject. I teach students to get prepared for college entry level exam. I also use to teach undergraduate students and guide them through their career aim.
5.00+
2+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Related Video
In this video, A mixture of methanol and air in a large polycarbonate bottle is ignited. The resulting rapid combustion reaction, often accompanied by a dramatic ‘whoosh’ sound and flames, demonstrates the large amount of chemical energy released in the combustion of alcohol
Students also viewed these Sciences questions
-
Using ethanol as your only source of carbon atoms, design a synthesis for the following compound:
-
Using acetylene as your only source of carbon atoms, design a synthesis of trans-5- decene:
-
Using acetylene as your only source of carbon atoms, design a synthesis of cis-3- decene:
-
Partners have fiduciary obligations to each other. Explain what this means and give examples.
-
Roadside Markets has a 6.75 percent coupon bond outstanding that matures in 10.5 years. The bond pays interest semiannually. What is the market price per bond if the face value is $1,000 and the...
-
Generate a trace for a program using software techniques. Use the trace to analyze the programs cache behavior.
-
Discuss the purpose of the Emergency Medical Treatment and Active Labor Act.
-
Betsy Ray started an accounting service on June 1, 20--, by investing $20,000. Her net income for the month was $10,000, and she withdrew $8,000. Prepare a statement of owners equity for the month of...
-
Consider SELECT statements listed below. SELECT L_PARTKEY, L_TAX, COUNT(*) FROM LINEITEM GROUP BY L PARTKEY, L_TAX; SELECT O TOTALPRICE, COUNT(*) FROM ORDERS GROUP BY O TOTALPRICE; SELECT L_TAX,...
-
As operations manager of Holz Furniture, you must make a decision about adding a line of rustic furniture. In discussing the possibilities with your sales manager, Steve Gilbert, you decide that...
-
Refer to Example 7.4. For this problem the correlation matrix is as follows: a. Since the zero-order correlations are very high, there must be serious multicollinearity. Comment. b. Would you drop...
-
Stepwise regression. In deciding on the best set of explanatory variables for a regression model, researchers often follow the method of stepwise regression. In this method one proceeds either by...
-
Nicks Novelties, Inc., is considering the purchase of electronic pinball machines to place in amusement houses. The machines would cost a total of $300,000, have an eight-year useful life, and have a...
-
Write a system of equations that has the given augmented matrix. a. \(\left[\begin{array}{lll:l}6 & 7 & 8 & 3 \\ 1 & 2 & 3 & 4 \\ 0 & 1 & 3 & 4\end{array} ight] \) b. \(\left[\begin{array}{lll:l}1 &...
-
Write each system in augmented matrix form. a. \(\left\{\begin{array}{l}4 x+5 y=-16 \\ 3 x+2 y=5\end{array}\\ ight.\) b. \(\left\{\begin{array}{l}x+y+z=4 \\ 3 x+2 y+z=7 \\ x-3 y+2 z=0\end{array}...
-
Write \([\mathrm{A}][\mathrm{X}]=[\mathrm{B}]\), if possible, for the matrices given in Problems 17-18. \([\mathrm{A}]=\left[\begin{array}{rrr}4 & 1 & 0 \\ 3 & -1 & 2 \\ 2 & 3 & 1\end{array}...
-
To manufacture a certain alloy wheel, it is necessary to use \(33 \mathrm{oz}\) of metal A and \(56 \mathrm{oz}\) of metal B. It is easier for the manufacturer to buy and mix two products that come...
-
Clint Eastwood is fifteen years older than fellow actor Goldie Hawn. If the sum of their birth years is 3,875 , in what year was Hawn born?
-
If you live where there is snow, do as Benjamin Franklin did more than 200 years ago, lay samples of light and dark cloth on the snow and note the differences among the rates of melting beneath the...
-
A new car sold for $31,000. If the vehicle loses 15% of its value each year, how much will it be worth after 10 years?
-
Rationalize each of the reactions in Fig. P21.56 with a mechanism, using the curved-arrow notation where possible. In part (d), identify compound A and show the mechanism for its formation. (Do not...
-
The reaction of Grignard reagents with nitriles is another method of preparing ketones. The example of this synthesis is shown in Fig. P21.57. Identify compound A, and give a mechanism for its...
-
How would you differentiate between the compounds in each of the following pairs? (a) p-ethylbenzoic acid and ethyl benzoate by IR spectroscopy (b) N-methylpropanamide and N-ethyl acetamide by proton...
-
What percentage of the American opportunity tax credit may be refundable?
-
You want to buy a house. You have $50,000 saved for the down payment and can pay $2,000 per month on the mortgage. The 30-year mortgage interest rate is 9% per year (equal to 0.75% per month). What...
-
Compute the Social Security and Medicare for Paul, Alex, and Andrea. The rates are 6 . 2 % for Social Security and 1 . 4 5 % for Medicare. Assume the Social Security limit is $ 1 4 2 , 8 0 0 and the...

Study smarter with the SolutionInn App


