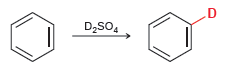
When benzene is treated with D 2 SO 4 , a deuterium atom replaces one of the
Question:
Transcribed Image Text:
D2SO,
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 55% (9 reviews)
H b8...View the full answer

Answered By

Ankit Mahajan
I am an electrical engineering graduate from Thapar institute of engineering and technology.
Qualified exams - GATE 2019,2020.
CAT EXAM 2021- 91.4 percentile
SSC EXAMS- 2019,2020,2021
AFCAT EXAM- 2019,2020,2021
I want to share my knowledge with other people so that they can achieve the same.
I have strong hold Mathematics, Electrical engineering and all the subjects related.
Just give me a problem and I will give you the solution of it.
5.00+
1+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
When benzene is treated with D2SO4, deuterium slowly replaces all six hydrogens in the aromatic ring. Explain.
-
When benzene is treated with propene and sulfuric acid (see structures of reactants below), two different monoalkylation products are possible. Draw their structures. Which one do you expect to be...
-
When benzene is treated with excess D2SO4 at room temperature, the hydrogens on the benzene ring are gradually replaced by deuterium. Write a mechanism that explains this observation.
-
Migration is a popular strategy among many species. Monarch butterflies migrate between the Sierra Madre mountains in Mexico and many locations across the USA and Canada. Answer the following...
-
Should a small business owner always purchase the products with the lowest prices? Why or why not?
-
As you've seen in this chapter, activity based costing (ABC) systems are useful in helping companies make better decisions about pricing, product mix, and cost management related to product design...
-
Trial weight runs have been completed, and the final weight calls for approximately 150 g. Using -inch plate steel, what size piece should be cut if a -inch welding rod is to be used for attaching...
-
The fill amount of bottles of a soft drink is normally distributed, with a mean of 2.0 liters and a standard deviation of 0.05 liter. If you select a random sample of 25 bottles, what is the...
-
Costs per Equivalent Unit and Production Costsof direct materials.Cost per equivalent units of $ 9 . 6 0 for Direct Materials and $ 3 . 0 0 for Conversion Costs.a . Cost of beginning work in process...
-
Slater & Gordon (S&G) became the worlds first listed law firm in 2007. The company is headquartered in Melbourne, where it was founded in 1935, by William Slater and Hugh Gordon....
-
Calculate A o R and G o R for the reaction C 6 H 6 (l) + 15/2O 2 (g) 6CO 2 (g) + 3H 2 O(l) at 298 K from the combustion enthalpy of benzene and the entropies of the reactants and products.
-
A specialty fabrication shop has the following assembly processes for one of the products it makes: Process 1 feeds a completed component to Process 2; Process 2 feeds a completed assembly to Process...
-
For each of the systems in Exercise 1.1.1. write down the coefficient matrix A and the vectors x and b. Exercise 1.1.1 Solve the following systems of linear equations by reducing to triangular form...
-
Exhibiting a note to its maker and demanding payment. a. bearer ins trument b. blank indors ement c. counterclaim d. dishonorment e. holder in due course f. indorsee g. indorsement h. indorser i....
-
The deduction of amounts owed claimed by the maker of a note or the drawer of a draft from the amount demanded by an immediate party. a. bearer ins trument b. blank indors ement c. counterclaim d....
-
A claim that states that the instrument was void from the beginning. a. bearer ins trument b. blank indors ement c. counterclaim d. dishonorment e. holder in due course f. indorsee g. indorsement h....
-
A claim for not paying commercial paper that relates to the circumstances leading to the issuing of the paper rather than to the paper itself. a. bearer ins trument b. blank indors ement c....
-
A person who has accepted commercial paper in good faith, for value, before maturity, and without notice of any defects in the instrument. a. bearer ins trument b. blank indors ement c. counterclaim...
-
For each polynomial function, one zero is given. Find all other zeros. (x) = 4x 3 + 6x 2 - 2x - 1; 1/2
-
Interest Compounded Annually. When P dollars is invested at interest rate i, compounded annually, for t years, the investment grows to A dollars, where A = P(1 + i) t . Trevor's parents deposit $7800...
-
(a) Two isomeric S"2 products are possible when sodium thiosulfate is allowed to react with one equivalent of methyl iodide in methanol solution. Give the structures of the two oroducts. ...
-
When methyl iodide at 0.I M concentration is allowed to react with sodium ethoxide at 0.1 M concentration in ethanol solution, the product ethyl methyl ether is obtained in good yield. Explain why...
-
When methyl iodide at 0.I M concentration is allowed to react with sodium ethoxide at 0.1 M concentration in ethanol solution, the product ethyl methyl ether is obtained in good yield. Explain why...
-
You observe the price of a latte changes from $4 to $5. Instructions: Round your answer to one decimal place. If you are entering a negative number include a minus sign. a. What is the percentage...
-
Layer 2 switches work at a simple level for communication and switching. With technology advancing and routers being able to make more complex decisions, why is this method still used?
-
Explore the significance of intermedial allusions in multimedia storytelling, such as in graphic novels or hypertext fiction, and their implications for narrative coherence and interpretation.

Study smarter with the SolutionInn App


