When trans-1-phenylpropene is treated with bromine, some syn addition is observed. Explain why the presence of a
Question:
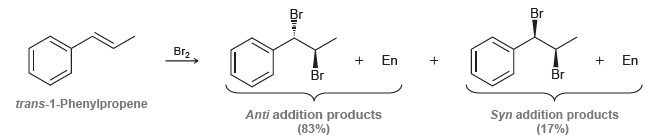
Transcribed Image Text:
Br + En Br2 En Br Br Syn addition products (17%) trans-1-Phenylpropene Anti addition products (83%)
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 75% (8 reviews)
The bromonium ion can open before a brom...View the full answer

Answered By

Susan Juma
I'm available and reachable 24/7. I have high experience in helping students with their assignments, proposals, and dissertations. Most importantly, I'm a professional accountant and I can handle all kinds of accounting and finance problems.
4.40+
15+ Reviews
45+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
When 2-methylpropane is treated with bromine in the presence of UV light, one product predominates. (a) Identify the structure of the product. (b) Draw the structure of the expected minor product....
-
Consider the structure of the following compound: (a) When this compound is treated with bromine under conditions that favor monobromination, two stereoisomeric products are obtained. Draw them, and...
-
Compound A has molecular formula C 5 H 11 Br. When compound A is treated with bromine in the presence of UV light, the major product is 2,2-dibromopentane. Treatmentof compound A with NaSH (a strong...
-
6.33. The "intrinsic gain" of a MOSFET oper- ating in saturation is defined as gmro. De- rive an expression for gmro and plot the result as a function of ID. Assume VDs is constant.
-
What is the optimum line of sight?
-
Daniel deposits \($20\),000 into an account earning interest at 6 percent per year compounded monthly. He wishes to withdraw \($1\),200 at the end of each quarter. For how many quarters can he make...
-
Suppose you are a salesperson working 50 weeks per year, five days a week, eight hours a day. You want to make \($50,000\) per year, which is based on a 10 percent commission of gross sales. How many...
-
Vanishing Games Corporation (VGC) operates a massively multiplayer online game, charging players a monthly subscription of $ 15. At the start of January 2015, VGCs income statement accounts had zero...
-
How do mentorship frameworks accommodate the nuanced interplay of individual aspirations, cultural contexts, and socio-economic landscapes, facilitating the cultivation of diverse talent pools with...
-
Explain the challenges faced by managerial accountants in effectively communicating managerial accounting information to various interested users.
-
Identify the product of the following reaction: Br Br ONa 2 NaBr C4H3O2 Nao
-
For each of the following cases, read the curved arrows and identify which arrow-pushing pattern is utilized: a. b. c. d. e. f. g. h. i. :Ci: -0-H - 0-H 0:0-I :O:
-
Corners of equal size are cut from a square with sides of length 8 meters (see figure). (a) Write the area A of the resulting figure as a function of x. Determine the domain of the function. (b) Use...
-
"How does a company's liquidity position impact its ability to meet short-term obligations and maintain financial stability?"
-
Lets compare a savings plan that pays 6% simple interest versus another plan that pays 6% annual interest compounded quarterly. If we deposit $8,000 into each savings account, how much money will we...
-
Ace Industries borrowed $230,000 amortized over 10 years at a rate of j12=6% with monthly payments (rounded up to the next cent). Calculate their final payment.
-
How can diversifying credit types, such as having a mix of credit cards, loans, and a mortgage, positively impact one's credit score and overall credit management?
-
A florist determines the probabilities for the number of flower arrangements they deliver each pa number in the boom Part 1: 4 points day. Part 2: 4 points Part 3: 2 points D) Part 4: 2 points 12...
-
To discourage tobacco use, federal law required all cigarette packages manufactured or sold in the United States to bear color graphics depicting the negative health consequences of smoking. The...
-
ABC company leased new advanced computer equipment to STU Ltd on 1 January 2019.STULtd has to pay annual rental of $290,000 starting at 1 January 2019. It is a four years lease with ultimate rental...
-
Explain whether the electron arrangement for these atoms is the ground state or an excitedstate: a) Energy 3 Nitrogen b) Energy 2p 5 4 Carbon
-
Show an energy level diagram for the MOs for He 2 and show how the electrons would be arranged in these MOs.
-
Indicate the type of atomic orbitals that are overlapping to form each of the different kinds of bonds in CH3OCH3 (For example, a carbon sp3 AO and a hydrogen Is AO). What kinds of orbitals are...
-
What percentage of the American opportunity tax credit may be refundable?
-
You want to buy a house. You have $50,000 saved for the down payment and can pay $2,000 per month on the mortgage. The 30-year mortgage interest rate is 9% per year (equal to 0.75% per month). What...
-
Compute the Social Security and Medicare for Paul, Alex, and Andrea. The rates are 6 . 2 % for Social Security and 1 . 4 5 % for Medicare. Assume the Social Security limit is $ 1 4 2 , 8 0 0 and the...

Study smarter with the SolutionInn App


