Predict the major product(s) for each of the following reactions: 1) MCPBA 1) , THF 2),, NaOH
Question:
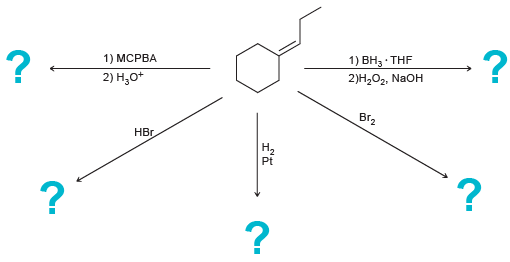
Transcribed Image Text:
1) MCPBA 1) ВН, THF 2)Н,О, NaOH 2) Н,о* ВГz НЕ. На Pt
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 68% (16 reviews)
HO OH En Br 1 M...View the full answer

Answered By

Hamza Amjad
Currently I am student in master degree program.from last two year I am tutring in Academy and I tought many O/A level student in home tution.
4.80+
3+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
Predict the major product (or products) formed when each of the following reacts with a mixture of concentrated HNO3 and H2SO4. (a) (b) (c) 4-Chlorobenzoic acid (d) 3-Chlorobenzoic acid (e)...
-
Predict the major product from each of the following reactions. (a) (b) (c) (d) (e) (f) (g) (h) (i) CH NH2, cat. HA N-H, cat. HA NH2 cat. HA PPha (1) HS SH (2) Raney Ni, H2 0 CH2PPha (excess) O. O...
-
Predict the major product from each of the following reactions. (a) (b) (c) (d) (e) HO OH NH2 HA (cat.) (1) HCN (2) LiAIH4 (3) H2o mCPBA
-
Assume that 3-month Treasury bills totaling $23 billion were sold in $10,000 denominations at a discount rate of 5.200%. In addition, the Treasury Department sold 6-month bills totaling $21 billion...
-
Develop and prototype a new interface design for the system's function using Graphical User Interface. Include common interface functions such as (textboxes, radio button, drop-down menus, check...
-
Air enters the compressor of a simple gas turbine at a pressure of \(1 \mathrm{bar}\) and a temperature of \(25{ }^{\circ} \mathrm{C}\). The compressor has a pressure ratio of 15 , and an isentropic...
-
A large MNE in the cookware industry was having difficulties maintaining its market share due to a number of mergers among other competing firms in the industry. The MNE, with corporate headquarters...
-
In the Olympiad of 708B.C, some athletes competing in the standing long jump used handheld weights called halters to lengthen their jumps (Figure). The weights were swung up in front just before...
-
The client's contract called for MSI to conduct 1000 interviews under the following quota guidelines: 1. Interview at least 400 households with children. 2. Interview at least 400 households without...
-
Richard chooses technique 0 and 2 requiring 10+10-20 efforts and provising 10+11=21 benefits. Hence, 21 is returned as the output Example 2: input1: 3 input2: (10,10,10,10) input3: (10,11,12,15)...
-
Would water be a suitable proton source to protonate the following compound? ONa
-
When 3-bromo-3-ethylpentane is treated with sodium acetylide, the major products are 3-ethyl-2-pentene and acetylene. Explain why the carbon skeleton does not change in this case, and justify the...
-
What does it mean to license a brand? What is co-branding?
-
Will internationalisation strategies of companies improve the domestic country competitiveness?
-
An economist estimated the learning curve for the production of Tesla vehicles to be: AC = 433,094*N -0.135 where AC = average production cost and N = cumulative production. Data used was annual....
-
What was the percentage change in the number of TANF families between 1996 and 2008? What are some possible explanations for this change? What was the number of TANF families in 2008? For the most...
-
Answer each of the questions and briefly explain your reasoning. a) Describe the difference between patents and design registration. Do they need to be registered? (4 marks) b) In what circumstances...
-
Consider an agent whose behavior is best described by expected utility. Calculate the value of the following prospect for the agent, assuming u(x) =x and initial wealth w = 1000 Payoff Probability...
-
The following information is available for two different types of businesses for the Year 1 accounting year. Diamond Consulting is a service business that provides consulting services to small...
-
Show, if u(x, y) and v(x, y) are harmonic functions, that u + v must be a harmonic function but that uv need not be a harmonic function. Is e"e" a harmonic function?
-
Explain why the reaction of the cis-isomer of this compound with potassium tert-but oxide in tert-butanol is about 500 times faster than that of thetrans-isomer. Br C(CH3)3
-
Explain which of these compounds has a faster rate of E2elimination: CH CH3 CI 'CI
-
Frequently, several different routes can potentially be used to synthesize a desired compound. For example, the following two routes can be envisioned for the preparation of cyclopentyl methyl ether....
-
Your GST bill has arrived. You need to pay GST on $153,890. How much do you need to pay in GST? Explain.
-
Given the fact that the telecom industry is extremely competitive, is it likely that Empire will find willing benchmarking partners among it's competitors?
-
Moxie Inc., a company that produces typewriter replicas, has fixed costs of $20,000 and variable costs of $18 per unit of output. Their expected unit sales is 10,000 units. What is the unit cost of...

Study smarter with the SolutionInn App


