A first-order decomposition reaction is observed to have the following rate constants at the indicated temperatures. Estimate
Question:
A first-order decomposition reaction is observed to have the following rate constants at the indicated temperatures. Estimate the activation energy.
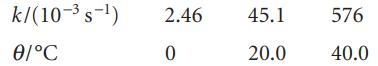
Transcribed Image Text:
k/(10-s-) Ꮎ/°C 2.46 0 45.1 20.0 576 40.0
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 60% (5 reviews)
we have K AeEaRT so lnK2K1 E...View the full answer

Answered By

Marvine Ekina
Marvine Ekina
Dedicated and experienced Academic Tutor with a proven track record for helping students to improve their academic performance. Adept at evaluating students and creating learning plans based on their strengths and weaknesses. Bringing forth a devotion to education and helping others to achieve their academic and life goals.
PERSONAL INFORMATION
Address: , ,
Nationality:
Driving License:
Hobbies: reading
SKILLS
????? Problem Solving Skills
????? Predictive Modeling
????? Customer Service Skills
????? Creative Problem Solving Skills
????? Strong Analytical Skills
????? Project Management Skills
????? Multitasking Skills
????? Leadership Skills
????? Curriculum Development
????? Excellent Communication Skills
????? SAT Prep
????? Knowledge of Educational Philosophies
????? Informal and Formal Assessments
0.00
0 Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
A first -order decomposition reaction is observed to have the following rate constants at the indicated temperatures. Estimate the activation energy. k/(10-3 s-1) 2.46 45.1 576 0/C 20.0 40.0
-
A charged particle is observed to have a total energy of 0.638 MeV when it is moving at 0.600c. If this particle enters a linear accelerator and its speed is increased to 0.980c, what is the new...
-
A 1.00-kg object is observed to have an acceleration of 10.0 m/s2 in a direction 30.0° north of east (Fig. P5.23). The force F2 acting on the object has a magnitude of 5.00 N and is directed...
-
Harry Bhel carries a business as a sole proprietorship. During its 2022 fiscal period, its first year of operations, the business had cash sales of $123,000. It also has sales on account of $46,000,...
-
A company produces scooters used by small businesses, such as pizza parlors, that find them convenient for making short deliveries. The company is notified whenever a scooter breaks down, and the...
-
An asset used in a 4-year project falls in the 5-year MACRS class for tax purposes. The asset has an acquisition cost of $7.6 million and will be sold for $1.4 million at the end of the project. If...
-
Consider PowerBar, the strategy for which is summarized in the PowerBar case. What implications for the culture, structure, systems, and people would you suggest given the nature of the product and...
-
On June 10, Diaz Company purchased $8,000 of merchandise from Taylor Company, FOB shipping point, terms 2/10, n/30. Diaz pays the freight costs of $400 on June 11. Damaged goods totaling $300 are...
-
A system consisting of a small 1.20-kg object attached to a Ax(cm) light spring oscillates on a smooth, horizontal surface. A 5.00- graph of the position x of the object as a function of time is 4.00...
-
An American firm has the opportunity to acquire a target company in Mexico. The firm anticipates owning the subsidiary for 8 years. Is this a profitable investment, and if so, what is the projected...
-
The rate law for the reaction in Exercise 22.2a was reported as d[C]/dt = k[A][B][C]. Express the rate law in terms of the reaction rate; what are the units for k in each case? Data in Exercise 22.2a...
-
The gas-phase decomposition of acetic acid at 1189 K proceeds by way of two parallel reactions: What is the maximum percentage yield of the ketene CH 2 CO obtainable at this temperature? (1) CH3COOH...
-
Show that the two formulas for x2 on pages 368 and 369 are equivalent.
-
In August 2010, the Federal Reserve announced that as the mortgage-backed securities it owns matured, it would reinvest the funds by buying U.S. Treasury securities. How would these actions affect...
-
An article in the Wall Street Journal discussed put options on Facebooks stock. One put option had a strike price of $25. The article observed: Puts are contracts that give the buyer the right to...
-
In the fall of 2012, an article in the New York Times speculated that the Federal Reserve could announce a new round of asset purchases, expanding its balance sheet for the third time since 2008. a....
-
Suppose that Bank of America lends $100,000 to Jills Jerseys. Using T-accounts, show how this transaction is recorded on the banks balance sheet. If Jills spends the money to buy materials from Zachs...
-
Suppose that the Dow Jones Industrial Average is above the 13,000 level. If the Dow were to fall to 10,000, who would gain the most: investors who had bought call options, investors who had sold call...
-
There are a very large number of mutual funds from which an investor can choose. Each mutual fund has its own mix of different types of investments. The data BestFunds1 present the one-year return...
-
Marc Company assembles products from a group of interconnecting parts. The company produces some of the parts and buys some from outside vendors. The vendor for Part X has just increased its price by...
-
Discuss the features, advantages, and limitations of the Michaelis-Menten mechanism of enzyme action.
-
Distinguish between the primary quantum yield and overall quantum yield of a chemical reaction. Describe an experimental procedure for the determination of the quantum yield.
-
Summarize the main features of the Forster theory of resonance energy transfer. Then, discuss FRET in terms of Forster theory.
-
19 Solve the equation for the requested variables. b(dy + z) =e+v Solve for b: b = Solve for y: y = entry tip: if you have several parts to a numerator or denominator, put them in parenthesis. To 2x...
-
2. An analysis and aging of Oriole Corp. accounts receivable at December 31, 2025, disclosed the following. Amounts estimated to be uncollectible $182,300 Accounts receivable 1,786,000 Allowance for...
-
Alex and Addison are married and have the following income items: Alex's salary Addison's Schedule C net profit Interest income $ 64,750 45,200 2,500 Addison's self-employment tax was $6,387....

Study smarter with the SolutionInn App


