The gas-phase decomposition of acetic acid at 1189 K proceeds by way of two parallel reactions: What
Question:
The gas-phase decomposition of acetic acid at 1189 K proceeds by way of two parallel reactions:
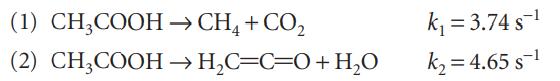
What is the maximum percentage yield of the ketene CH2CO obtainable at this temperature?
Transcribed Image Text:
(1) CH3COOH (2) CH3COOH → CH4 + CO₂ → H₂C=C=O + H₂O k₁ = 3.74 s ¹ k₂=4.65 s-¹
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 33% (3 reviews)

Answered By

Anurag Agrawal
I am a highly enthusiastic person who likes to explain concepts in simplified language. Be it in my job role as a manager of 4 people or when I used to take classes for specially able kids at our university. I did this continuously for 3 years and my god, that was so fulfilling. Sometimes I've skipped my own classes just to teach these kids and help them get their fair share of opportunities, which they would have missed out on. This was the key driver for me during that time. But since I've joined my job I wasn't able to make time for my passion of teaching due to hectic schedules. But now I've made a commitment to teach for at least an hour a day.
I am highly proficient in school level math and science and reasonably good for college level. In addition to this I am especially interested in courses related to finance and economics. In quest to learn I recently gave the CFA level 1 in Dec 19, hopefully I'll clear it. Finger's crossed :)
4.80+
2+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
Estimate the liquid diffusivity of acetic acid at 25oC in a dilute solution of:(a) Benzene,(b) Acetone,(c) Ethyl acetate, and(d) Water by an appropriate method. Compare the estimated values with the...
-
Find the viscosity of Acetic acid at 300K.
-
Calculate the volume of 47 g of acetic acid at 20 C ( = 1.05 g / mL).
-
How do recruitment and selection practices contribute to high performance in an organization?
-
PricewaterhouseCoopers Saratoga, in its 2005/2006 Human Capital Index Report, indicated the average number of days it took for an American company to fill a job vacancy in 2004 was 48 days. Sample...
-
Consider the following project for Hand Clapper, Inc. The company is considering a 4-year project to manufacture clap-command garage door openers. This project requires an initial investment of $18...
-
The Korvette concept was started and run by one person and his group of friends. How could its failure have been avoided? Was the problem one of strategy (overexpansion), or was it organizational?...
-
Jorgensen Corporation uses standard costs with its job order cost accounting system. In January, an order (Job No. 12) for 1,900 units of Product B was received. The standard cost of one unit of...
-
A 480-g iron pan is heated on a stove to a temperature of 130 C. The pan is then dropped into a partially-filled sink of water at 20 C. The final temperature reached by the sink and the pan is 40 C....
-
A sweatshirt supplier is trying to decide how many sweatshirts to print for the upcoming NCAA basketball championships. The final four teams have emerged from the quarterfinal round, and there is now...
-
A first-order decomposition reaction is observed to have the following rate constants at the indicated temperatures. Estimate the activation energy. k/(10-s-) /C 2.46 0 45.1 20.0 576 40.0
-
The following data have been obtained for the decomposition of N 2 O 5 (g) at 67C according to the reaction 2 N 2 O 5 (g) 4 NO 2 (g) + O 2 (g). Determine the order of the reaction, the rate...
-
You have been asked to put together a capacity plan for a critical operation at the Surefoot Sandal Company. Your capacity measure is number of machines. Three products (men's, women's, and...
-
An article in the Wall Street Journal noted that, Options traders appeared to be taking a bullish approach to Target because during the previous day they purchased 68,000 calls on the companys stock,...
-
If the exchange rate between the yen and the dollar changes from 75 = $1 to 90 = $1, is this good news for Sony? Is it good news for U.S. consumers? Is it good news for U.S. firms that export to...
-
If $2 buys 1 and 2.2 buys 1, how many euros are required to buy $1?
-
A student makes the following observation: It currently takes 80 yen to buy 1 U.S. dollar, which shows that the United States must be a much wealthier country than Japan. But it takes more than 1...
-
In a column in the New York Times, Uwe Reinhardt, an economist at Princeton, compared health care spending in Germany and the United States: Total national health spending in Germany. . . amounted to...
-
The manufacturer of Boston and Vermont asphalt shingles provides its customers with a 20-year warranty on most of its products. To determine whether a shingle will last as long as the warranty...
-
Outline some of the major problems confronting an international advertiser.
-
On the basis of the following proposed mechanism, account for the experimental fact that the rate law for the decomposition 2 N2O5 (g) → 4 NO2 (g) + O2 (g) is v=k [N205]' k,ki (I) NO NO.+NO, (2)...
-
Consider the following mechanism for the thermal decomposition ofR2: Where R2, PA' PB are stable hydrocarbons and Rand R' are radicals. Find the dependence of the rate of decomposition of R, on the...
-
Refer to Fig. 23.3 and determine the pressure range for a branching chain explosion in the hydrogen-oxygen reaction at (a) 700 K, (b) 900 K.
-
Morganton Company makes one product and it provided the following information to help prepare the master budget: a. The budgeted selling price per unit is $70. Budgeted unit sales for June, July,...
-
k On January 1, 2024, the Marjlee Company began construction of an office building to be used as its corporate headquarters. The building was completed early in 2025. Construction expenditures for...
-
Dividends per share Seventy-Two Inc., a developer of radiology equipment, has stock outstanding as follows: 80,000 shares of cumulative preferred 3% stock, $20 par and 410,000 shares of $25 par...

Study smarter with the SolutionInn App


